I have covered hair regeneration research by Yokohama National University (Japan) based scientist Dr. Junji Fukuda a few times in the past. His team’s most important work relates to hair multiplication via the production of hair follicle germs.
However, Dr. Fukuda achieved global notoriety in 2018 due to the French Fries chemical related hair loss “cure”. In recent years, the Yokohama Team has published numerous papers. Moreover, they plan to begin hair multiplication trials in humans in 2023.
I first wrote this post in 2021, and will update it regularly. This is due to the importance that I attach to Dr. Fukuda’s groundbreaking work. Make sure to also check Fukuda Lab for the latest news. They seem to publish a new study every few months. Also see the hair section of their website.
April 12, 2023
Yokohama Researcher Junji Fukuda’s New Interview
A detailed new interview with Dr. Junji Fukuda (h/t “Ben”). Nothing in there about their human trial plans for 2023, but nevertheless very interesting.
They are pursuing three different mechanisms for hair regeneration:
- Reactivating the hair follicle tissue that remains in the scalp via transplant special cells. These cells give instructions on reactivation of dormant hairs as well as increasing the thickness of existing hair. For this method, they are working together with Rohto Pharmaceutical. Note that he expects human clinical trials for this method to begin “within the next few years”.
- Inject (transplant) cell tissue prepared in vitro into the scalp and induce new hair follicle tissue to grow. This is based on their successful 2018 results related to “hair follicle primordia” that I discussed in the earlier hair follicle germs link.
- Create and transplant hair follicle tissue containing hair from cells through culture. Based on their 2022 work in relation to “hair follicle organoid”. I discussed this in this very post in the prior updates further below.
In is interesting how much the students are involved in this research. Dr. Fukuda even gives credit to the last method above to one his student’s discoveries. Something that almost never seems to happen in the western world. Even the new company Trichoseeds is part owned by his student lab researchers:
“I founded a venture company together with the students in my laboratory so that the technology could be used for the practical application of hair regenerative medicine. I started the project because I thought that students would be more motivated if they were doing research for social implementation, so they would be able to benefit from it as well.”
October 21, 2022
The un-finalized paper that I mentioned in my prior June update was just released today. Lots of media coverage.
In yet another breakthrough, the Yokahama University team led by Junji Fukuda and Tatsuto Kageyama have managed to grow mature mice hair follicles in cultures. Actual paper here and was published today. The news is already being covered widely in a number of publications, including US News and Yahoo.
According to Newscientist:
Mature hair follicles have been grown in a laboratory for the first time, in a move that could one day treat hair loss.
The hair follicles grew for up to one month, reaching 3 millimetres length. According to Dr. Fukuda, this is probably due to the fact that the mice hair cycle is about one month. Quote from the good doctor:
Our next step is to use cells from human origin, and apply for drug development and regenerative medicine.
Also note that the same team released a new paper on October 1 titled: “Impacts of manipulating cell sorting on in vitro hair follicle regeneration.”
June 25, 2022
The updates from Dr. Fukuda and his team at Yokohama National University (YNU) keep coming and I am excited. Thanks to “Ben” for the links to the two latest papers. Also make sure to see Dr. Fukuda’s patents page.
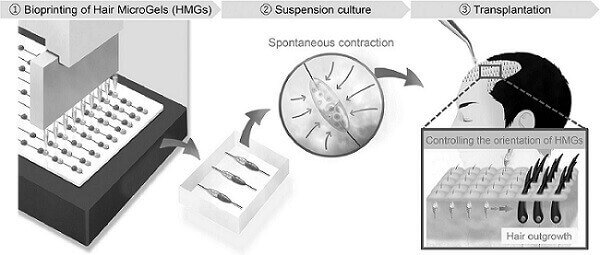
Junji Fukuda and TrichoSeeds
- On June 16, the Fukuda Lab published a new paper titled: “Bioprinting of hair follicle germs for hair regenerative medicine.” This study outlines an approach for the scalable and automated preparation of highly hair-inductive grafts using a bioprinter (image above). The study link also includes two interesting videos at the bottom.
However, perhaps of most interest to me, they declare in the paper that:
“Ayaka Nanmo, Tatsuto Kageyama and Junji Fukuda are co-founders of TrichoSeeds, a company that provides hair regeneration medicine.”
This confirms the original YNU announcement about TrichoSeeds from earlier this year that a number of readers discussed.
- Also published in June 2022 (but not yet peer-reviewed), the Fukuda and Kageyama team authored a paper titled: “Reprogramming of three-dimensional microenvironments for in vitro hair follicle induction.”
While doing research for this update, I discovered the below new April 2022 video published by Yokohama National University in regards to Dr. Fukuda. This all looks to be the real deal….although I said the same in the past about the Dr. Tsuji/Riken team. Also see this earlier 2017 video presentation from Dr. Fukuda.
Update: April 15, 2022
Yokohama University and Dr. Junji Fukuda
- In April 2022, Dr. Fukuda and his team published a paper titled “Effects of the PI3K/Akt signaling pathway on the hair inductivity of human dermal papilla cells in hair beads.”
- In March 2022, the Yokohama team published an interesting new paper titled “Electrical stimulation to human dermal papilla cells for hair regenerative medicine.” I added it to my post on electricity and hair growth.
- Thanks to “Ben” for noticing that Yokohama University’s US patent application for its method for producing hair follicle germs got updated on February 10, 2022.
I consider Dr. Fukuda and his team to be among the five most important players in the world that are working on a hair loss cure. Per last year’s news (see further below), the Yokohama team plans to start human clinical trials in 2023.
In this latest research, the team managed to make new hair that was at first only growing underneath the skin of mice to sprout through the skin. They did this via the use of guide-inserted hair microgels (HMGs). They conclude:
“This approach is a promising strategy to advance hair regenerative medicine.”
Update: April 7, 2021
Interview with Dr. Tatsuto Kageyama
Yesterday, in our Discord chat, “DrPhil” posted a link to a new April interview with Dr. Tatsuto Kageyama of Yokohama National University. It is worth a full read.
After some quick research, I realized that Dr. Kageyama is a co-author in Dr. Fukuda’s important 2019 paper titled: “Preparation of hair beads and hair follicle germs for regenerative medicine.”
This latest interview with Dr. Kageyama is extremely insightful and encouraging.
Human Trials to Start in 2023
The most important news is that human trials will start in 2023. Moreover, the treatment will not be too costly, and it will be safe.
Some key quotes after translation:
“We started basic research using human cells in 2018, aiming to start clinical trials in 2023. And we are working hard together with the members of the laboratory so that we can deliver it to everyone as soon as possible.”
“We also believe that we will be able to resolve cost issues.”
Apparently, they are convinced that the cost of this treatment will be much lower in comparison to other regenerative medicine treatments. A nice change from Dr. Tsuji’s expensive hair loss cure.
The team’s mass culture technology does not necessitate expensive equipment. Moreover, the required amount of hair follicle primordia can be produced with a single culture container.
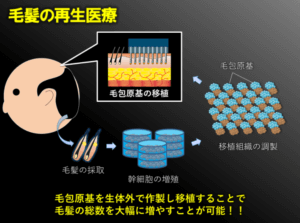
Hair Follicle Primordium Creation
The Yokohama National University team’s hair multiplication process involves creating hair follicle primordia. Epithelial cells and mesenchymal cells mix to form a tissue called “hair follicle primordium“. This is essentially the seed for hair growth.
The process this team uses entails:
- Extract a few healthy scalp hairs.
- Increase the “epithelial cells” and “mesenchymal cells” that exist in those follicles.
- Thereafter, cultivate a large amount of hair follicle primordia.
- Transplant these new cells to balding regions of the donor’s scalp to regenerate the hair.
It will be possible to produce thousands of hairs from several hairs. The whole process is considered to be safe due to the autologous nature of the treatment.
Japan and Regenerative Medicine
Best of all, in Japan, clinical trials in regenerative medicine are expected to move along faster than in the US or EU. The country has a rapidly aging society with low birth rates. Therefore, it is a world leader in anti-aging related research.
The important triumvirate of Riken (Tsuji), Shiseido, and Yokohama (Fukuda) are all based in Japan. I am very hopeful that at least one of these three will finally succeed in bringing a next generation hair loss treatment or cure to the market.
I’ll believe when I see it. As of now he is a tsuji until I see phase1 done with robust regrowth. Good to see other players in the game though.
Had a very successful FUE HT at ASMED in Turkey by Dr Erdogan. Give up on this pie in the sky tripe and go get it done. Remember this is ‘hairloss cure 2020’. 2020 has been and gone, we are years away from a real cure, only temporary fix is a HT boosted with propecia. Facts.
How many grafts and how much did it cost?
I live in Turkey,Noramlly it costs between 1000 to 3500 USD depends on your status,but great results İ’ve seen here.
Thank you as ever Admin for striking the tone of honest, patient optimism that is of great comfort to your readers. Cheers, with much gratitude.
Thanks! I want the cure for myself too :-)
I guess the problem is if you have been maintaining on anti androgens or 5alpha reductase inhibitors.. you will have to get off and lose hair 1st ro replace? Am I off base?
Who cares if it work. A moment of temporary baldness for permanent cure.
Still the question is how expensive it will be.
“We also believe that we will be able to resolve cost issues.”
Such good News!
Fortuna, The first questions are will it work and when will it be available. Skipping to the price part is a bit presumptuous :-)
I’m a little confused (probably just being thick). But this is hair cloning, right? If so, there is the cost of the cloning…then the cost of the surgery (multiple, if one has severe hair loss). Or am I missing something? The surgery (esp if it’s multiple) alone is expensive…but coupled with the cloning would be super costly I’d imagine.
Yea, it’s basically like hair cloning but not the same concept. It basically like… clone your hair but without genetic manipulation involved and other complex stuff that you would do on real clones. It’s called hair multiplication simply because it multiply your hair instead of cloning from A to Z.
Real cloning would be more expensive but the thing with hair multiplication is that some part of the hair might not be able to get multiplied with our current technology so maybe we’ll be able to clone 100x times the amount of hair that are immune to DHT and you’re basically cured FOR LIFE (but if we can multiply every part of the hair then it’s same result as hair cloning) while with real cloning technology we could clone an infinity number of hair with only 1 of your hair follicle. That’s what I understand about hair cloning and hair multiplication so far.
In summary: Hair multiplication might be able to multiply 1 hair follicle to 100 (probably enough to cover the rest of your body so you don’t have to worry about your amount of donor area) ones which is probably plenty enough for 99% of people and they would probably clone the hairs that are in the back of your scalp since they are DHT resistant. But for those who somehow don’t have hair in the back of their scalp but only 1 hair follicle left then they would need hair cloning.
@user12 well said
I’m not a health care professional. But cloning requires something to be grown in the womb, right? There are a lot of our body parts that are only grown in the womb, such as the inner ear hairs that we hear with, so I guess we need to either make humans a little less human-like, or artificial parts a little more human like. :D
I hope that age related baldness can be cure just like young age baldness … I’m 49 and with Fina and Minox I’m losing ground big time:( it started about 4 years ago, before then everything it was still ok
Lorence with hair multiplication you have an endless supply of hair at your disposal.
Best
Almost every men have male pattern baldness. It’s just that some get it younger and some get it older. This can definitely fix your baldness forever no matter the age since you have either female or male pattern baldness and the donor area which is the area in the back of your scalp are really strong and are also immune to DHT which means they can multiply it and put on top of your scalp and it would stay there for a life time.
49?
I wouldn’t feel too bad. Many men start losing it quite younger than that (i.e. early 20s).
ALL women eventually have some unwanted hair loss also, if they live to be past 50.
I’m still not happy with the timeline. Trials in 2023? So two years before they begin. Then a few more years to finish and analyze results. Then a few more years to…
We’re still looking at a decade here for a product. I guess it’s still good news, if it’s not another letdown.
Exactly !
That’s assuming they even start in time. I think more than a decade. Too late for me fellas, cue ball here I come
Bruh, don’t give up just yet… For now I recommend you use finasteride and other treatments for your hair to last as long as possible till the cure is available.
Lorence add a topical minox/finasteride that help me with a boost of growth and stabilization when the big 3 lost ground….I’m still losing ground but it seems to fluctuate. Some months look thicker while other months looks worse.
Topical Finasteride? How do you mean it?
I’ve summarized the information I could find about the Verteporfin research:
– The research paper [1] describes the mechanisms of scar formation in skin and a simple procedure to achieve full skin regeneration using a very safe FDA approved drug, Verteporfin. Full skin regeneration here refers to skin indistinguishable from normal skin with all the dermal appendages, including hair follicles. According to the paper, Verteporfin works by preventing a type of skin cell (ENF) from giving rise to another type of skin cell (EPF) that causes scarring during wound healing. The way it does this is by blocking mechanotransduction signaling, i.e. the ability of cells to sense mechanical forces.
– The studies were conducted on mice, but according to this article from The New York Times [2], it also works on pigs. Quoting from the article:
“The study involved mice, but the researchers, Dr. Michael Longaker, Stanford’s vice chair of surgery, and Geoffrey Gurtner, Stanford’s vice president of surgery for innovation, have now moved on to pigs, whose skin is closest to that of humans. With these new subjects, the surgeons made an incision as wide as a thumb and five inches long. When they sutured the cut and injected Verteporfin around the edge, there was dramatically less scarring.”
– The procedure on mice was, first, create a wound using a skin biopsy punch around 4mm in diameter. After wounding, inject 30 μL of Verteporfin (1 mg/mL) [3] around the edges of the wound. Relevant here is that repeated injections on day 4, 8 and 12 didn’t improve outcomes and had detrimental effects. A single injection after wounding worked best. The results were, control wounds: hairless scars; treated wounds: substantial hair growth by 30 days and indistinguishable from normal skin by 90 days.
– The drug used on patients is Visudyne. It is a 20 year old drug approved for the treatment of age-related macular degeneration and pathologic myopia. Each box contains a glass vial with 15 mg of Verteporfin powder and costs around 1000 USD. It is reconstituted by injecting 7mL of sterile water into the glass vial to provide 7.5mL of 2 mg/mL Verteporfin. The volume of reconstituted Verteporfin required to achieve 6 mg/m2 body surface area, around 5mL for an adult by Mosteller formula, is withdrawn from the vial and diluted with 5% Dextrose for Injection to a total infusion volume of 30 mL. It is administered intravenously over 10 minutes at a rate of 3 mL/minute. The second step is the light activation of Visudyne using a laser at 15 minutes after the start of the infusion, but this part is irrelevant for wound healing. Note that the dosage used on mice was minuscule in comparison at 30μL of 1 mg/mL Verteporfin, or 0.03mg of Verteporfin, and that it was injected in the skin instead.
– Patents on Verteporfin have all expired so anyone can manufacture it. There are manufacturers selling >99% purity Verteporfin at better prices for research purposes, but they don’t sell to patients.
– There is previous work [4] on the role that tension plays during wound healing on humans:
“embrace® Active Scar Defense is the only FDA-Cleared scar therapy system designed to relieve tension around incisions, general cuts and lacerations to prevent the formation of new, visible, raised scars before they start.”
Presumably, completely blocking mechanotransduction at the cellular level using a drug like Verteporfin would work much better.
Given that it works on mice and pigs. That there is a product that works on humans by reducing wound tension. And that wound healing mechanisms are well conserved among mammals. I would bet that it will work on humans. Very exiting. We need to raise awareness in the hair loss community to get trials started to answer whether it will regenerate terminal scalp hair. Hair loss won’t be the focus of most researchers and we could sit on a working cure for years otherwise.
[1] https://science.sciencemag.org/content/372/6540/eaba2374
[2] https://www.nytimes.com/2021/04/22/health/surgery-scar.html
[3] https://science.sciencemag.org/content/sci/suppl/2021/04/21/372.6540.eaba2374.DC1/aba2374_Mascharak_SM.pdf
[4] https://www.embracescartherapy.com/citations-and-patents
@toagi thank you!
Are you on the United States. The stuff does cause crazy hair growth. I’ve been taking it 23 days and I am usually a very non hairy man.
Hey! We’re working with a HT doctor to source and administer Verteporfin. Wondering if you’re interested in joining the discord to help.
I still remember too good how Tsuji will start trials in 2020. Which never happened.
Anyhow, Fukuda is the real deal.
StemsonTx trial announcement should be the next big news…hopefully!
Appreciate hearing positive news. We can’t ever expect things to happen quickly, but I certainly think a baldness cure will be available this decade. Knowing that can give us all a little extra confidence and something to look forward to :-) Until then, my subtle SMP added to my buzzed short hair transplants has me feeling a bit younger and less insecure. Still, nothing beats some lengthy hair growth, so I’ll look forward to that.
Hey … I’m actually looking to do this treatment – diffuse NW 5/6 and I have SMP – just curious, did the transplant add “realness” to the look for you? The examples I’ve seen look good, at least passable. I’m just worried when I go full slick that the SMP alone won’t be ideal (for me, for others, I support whatever works!!)
Lorence…I know you heard of topical finasteride minox combo topical. You are a frequent commenter here:) Anyway…look at MorrF or Minoxidil Max they have several compound minox solutions. There are a ton out there. I think the admin posted links to some in a previous post.
I’ve never heard of this but I’m going to look into it. Only thing I’m using now is topical minox from the store (5%).
Also the below, although I have never tried. Nor have I updated the post since 2016 it seems:
https://www.hairlosscure2020.com/extra-strength-minoxidil/
Thanks admin. I’ll take a look. I’m not big on topicals but I’ll give anything a try – if I see change then I’ll keep using (otherwise I scrap it). The combo stuff looks promising.
James it helped with a boost and stability. Far from a cure or anything like that but it helped me stabilize or slow the progression.
Good to know. Appreciate it. Can’t hurt to try.
I’ve been contemplating getting a HT – but with such little (thinning) donor hair it would be interesting to see what they’d say at a consult. There are a few docs out there that can work wonders though. We’ll see.
Hi James,
I’m in a similar position re donor hair. I had three consultations the year before last. Two said they could do something, but the third said I didn’t have enough hair to make it viable. I’ve decided to hang on in the hope there is some sort of hair multiplication in a few years time. So definitely worth getting at least three opinions.
Thanks. And yeah, the one that may say they can do something may just be out for the money and by something they mean very little. I don’t get why the donor hair is so thin. This is where it’s supposed to be fine! Ugh. If only I had thick sides I’d be in for multiple sessions.
The info on Yokohama is interesting. I think it’s something the young population can be hopeful for. Hopefully they wont pull a histogen or follica etc..I dont have much hair left and by the time the trials get finished the hair I have left will be long gone.
Tom you can be excited as well because as long as you have the cells required to make a HFG you are still in the game. Just because you don’t sprout hairs out of your hair, doesn’t mean you don’t have the tools required to Induce hair follicle neogenesis.
Have any of you ever tried to put 2-3 Finasteride tablets in Minoxidil? If so, with what results?
Let’s see what’s on the shelf within the decade? (If they survive?) Got a strong feeling it will still be HT’s, Fin, Min and Niz tho, seeing how slow things move. Hope to be surprised, but I wouldn’t bet your Bitcoins on it!
Why I feel this method resembles the TissUse or Replicel?
@lll. Hmmm interesting.
Hi Admin, just saw that Fukuda and Kageyama published a couple of new research papers regarding hair regeneration over the last months.
They‘ve been rather active I would say – they‘re onto something.
Thanks Ben! They seem very reputable. Below is sorted by date if anyone is interested:
https://pubmed.ncbi.nlm.nih.gov/?term=fukuda%2C+junji%5BAuthor%5D&sort=date
Thanks Ben. Nice contribution.
Lorence – I started topical dutasteride from strut with good success. Oral fin gave me sides but topical dut hasn’t. My skin can’t handle minox. I’m 1.5 months in so hopefully it stays that way. Haven’t regained much hair, if any, but my shedding basically stopped. Enough to keep me happy at least. M29
Have you considered asking a dermatologist to prescribe you oral minoxidil if topical irritates your skin?
I’m considering it, since I’m getting tired of the stickiness and residue from applying topicals.
I had fantastic results with oral dutasteride but had to stop due to sides and I’ve seen a gradual thinning since. I’d love to read an update on oral dut. So many different sellers & options have appeared this year.
Please write a detailed testimonial of your results in a comment to the below post if possible:
https://www.hairlosscure2020.com/dutasteride-avodart-hair-growth-success-and-failure-stories/
e.g., Did see results fast? How much regrowth did you experience in totally bald regions that had lost hair many years ago? What side effects did you get?
Thanks!
The attempts at hiding baldness probably started when mankind evolved from the animal that was similar to an ape, but not an actual ape. Anyway, how many millions of years have male humans been trying to deal with unwanted hair loss? It kind of makes me wonder, what the hell have researchers been doing for the past million/billion years? LOL!
Hi DH, congrats on the topical Dut success. I see you mentioned oral fin gave sides and topical Dut gives you zero sides. Can you tell me if you ever tried topical fin and if so, did it give you sides?
And where did you purchase your topical Dut? What is Strut?
I tried a topical fin + min but had to stop because I felt like the min was causing more shedding and dry scalp than helping anything. The topical dut I got is a gel that is much easier to apply and has really helped with my overly oily scalp. Strut is the website where I ordered it. Itskind of my last ditch effort to try to stabilize my shedding before I called it quits and just let it go.
Thank you admin my boy for this awesome wonderful news. All glory to the Lord Jesus Christ.
Woofy97! , Jan! what do you guys think of this ? Do you think they might be like tissuse or Replicel ?
Well, at least Jesus had hair-going by the old frescos. I doubt they would’ve painted him with a u-hawk! God it seems, wasn’t so kind when dolling out the hair genes to us mere mortals.
https://www.studyfinds.org/cure-baldness-protein-hair-growth/
Just bumped into this, sorry in advance if it has already been covered elsewhere on this page.
Covered here:
https://www.hairlosscure2020.com/stress-does-not-cause-permanent-hair-loss/
Mark I wouldn’t get too hyped about this guy he’s basically tsuji, just tsuji told us a specific year and this guy hasn’t and that upset people now people are hyped about this new player. I’ll believe it when it’s tested on human. These researchers need to walk the walk instead of talk the talk.
@woofy97 Thank you dude.
Hi DH, thanks for your response. I pondered trying topical Dut but I’m hesitant because of it’s long half life. Oral fin gave me bad sides and topical fin lowered my bp and gave bad chest pain. Low bp is an uncommon side effect of fin. My cardiologist immediately pointed it out to me when I told her about my experience.
Hey yall …. So, I am back in here after taking a year and a half off and I see nothing has changed. I have decided to go ahead with the Hair Transplant procedure. I am hoping to have it done by January. Also hoping that in 10-15 years when I may need another transplant that this whole hair loss thing is cured and I won’t need one.
I’m thinking of doing the same. Been Hanging round waiting for the cure right round for the last 10 years.
Question for Admin / Anyone Who Knows The Answer:
The more I look at “success” stories and pictures of people who had regrown hair, the only ones I find truly impressive are the ones where someone has been transitioning.
So, at the risk of stating the obvious or sounding like a Bro Scientist, that strongly suggests that the hormones are a key area that scientists should be focussing on.
I’ve read about a couple of drugs (like Spironolactone), and they seem to have good results with hair growth. The problem, unless you are transitioning, is that they can “castrate” you (not my words).
So, my question is this: Is there a middle ground? A drug that can help without affecting your sex hormones etc?
Dutasteride and Finasteride do give super results in some.
Anyone have diffuse thinning everywhere even in the donor area? Is this incredibly unusual?
I have been on rogaine and even avodart for years with no success. Ironically I added Nizoral consistently and this cut the shedding in half already so I’m not sure what that means.
I have no doubt there must be an AGA male latter on loss occurring but I suspect something else is also compounding this.
https://patents.justia.com/patent/20220041983
Patent published on February 10 in the US.
Would be great to get some information directly from Fukuda or Kageyama.
What about this stem cells treatment ?
Is it serious ? Does it work ?
https://www.vie-aesthetics.com/treatments/viestem-hair-with-regenera-activa/
https://www.hairlosscure2020.com/regenera-activa-amt-for-hair-loss/
Thank you for the article, but may I please ask you admin do you really think they will start human trials next year especially after the major earthquake that happened yesterday? And if so what timeline of a commercial release date?
I’ll drink another beer to that.. Thanks, Sir Admin. Let’s hope 2023 is the year. Until then, lots of FUE plus SMP plus very short buzz cut plus tan plus exercise will keep you looking young.
Why 2023 ???
I think we’re doomed no ?
Yep we are haha maybe we can get something from kintor the next years…..but the rest ll take 15 to 20 years….
Meanwhile HanBio is moving forward:
https://news.mtn.co.kr/news-detail/2022031710212379033
“With the approval of the cell treatment facility last year, the entry into preclinical trials, and the recent approval for the manufacturing of advanced biopharmaceuticals, we are standing at a very important start line for the fundamental and complete solution of the hair loss problem that Hanmo Bio is planning.”
That’s one bold statement. Looking forward to their results this (!) year.
I would bet the farm that Junji Fukuda, PhD is not going to do human trials next year!
More nonsense, I been hearing about potential cures for the past 45 years with only poor to no results from a couple products: minoxidil and finasteride. Until you see clear real result, not enhanced photos, don’t get excited. I cure will come some, day but 50 years from now is more realistic. The products you are talking about today are just more nonsense.
Great article – please email me if you need a volunteer aka a Guinea pig.
Admin
What do you think and the opinion of the group members in purchasing such products before the completion of the third clinical trials?
Pyrilutamide https://www.reddit.com/r/tressless/comments/vkcxjt/pyrilutamide_will_be_available_at_actifolic_in/?ref=share&ref_source=embed&utm_content=body&utm_medium=post_embed&utm_name=d5d095a3c3c3c2480067m_vmt_vxsource=b9
CB-03-01
https://actifolic.com/product/cb-03-01-solution/
I follow them on Discord, and some of our chat members are part of the group buys. Will let you know if I see any great before and after results.
Hate to say it, but heard it all before. More chance of being bitten in the arse by a rabid cabbage before you start sprouting hair like Java Man. Don’t throw out the pills and potions just yet kids.
I heard that if you get bitten in the arse by a rabid cabbage it grows hair!
I hear ya. I’d bet the house it would on some mice for sure.
Admin, Thank you as always. Interesting and great updates.
Hi admin – thanks for the update.
Unfortunately the video link does not work, can you repair it?
Hi Ben, one video link (where I say “this earlier 2017”) works for me. And the new April 2022 Fukuda video is embedded right below that link. Can you point out out which one is not working?
Sorry, all good now. It didn’t load before, it was maybe my own fault.
Very good news these are. I wonder how the RIKEN and KAKEN guys are connected?
Fukuda regularly references Tsuji et al in his papers. I hope he learns from their mistakes.
Fukuda‘s „Reprogramming of three-dimensional microenvironments for in vitro hair follicle induction.“ is similar to Stemson‘s and Epibiotech‘s plans – a human hair model to test new medicines and treatments. Or Tsuji‘s digital model ( https://www.riken.jp/en/news_pubs/research_news/rr/20211006_1/index.ht) from last year. These approaches should make R&D much faster.
According to this new Fukuda-paper they are also looking closer into the mechanisms of hair-greying – which sounds like they will likely research treatments for that as well. Exciting.
I lost hope on the Japanese, as they are hard worker but they don’t invent something from the scratch due to lack of creativity. The cure will come from USA.
@Ibrahem ….Exactly ! THE Japanese do not invent anything all new things come from America.
The Japanese create some things, look at the Yamanaka Factors. However remember the 1980’s: everyone thought they were going to take over the world because they stole Western ideas and made them more efficient. Never happened ofc. Hive minds. 90% of progress will continue to come out of the West this century.
We will see what comes of this. Not hyped anymore for hair cloning. Unless I can actually make an appointment with a hair transplant doctor doing this. Veterporfin.
Great news admin.
Moderator: All Caps shouting deleted.
Ok let me repeat myself in lower caps junji fukuda will most likely, not do human trials next year or ever! not in 20 years. It’s easier to get to the moon and start a colony then to clone a hair..
Facts
What about cloning hair on mars?
I know that’s why I was hoping elon musk would be able to set on mars by 2026 and start experiment’s but I don’t think he’ll get there until the late 2030s. Either way there’s little to no hope for a real hair loss cure which in my humble opinion can only be achieved through hair cloning.
I got an ad on my Instagram feed for this NJ compounding pharmacy. You can build your own compound. Just wanted to pass it along in case anyone is interested. I will be looking to get off the Fin pills this fall and I’m in the market for a compounded topical. I can’t vouch for these guys but I might try it.
https://www.cfspharmacy.pharmacy/hair-loss
Here in Europe cheap hair transplant centers are popping up like mushrooms after the rain. This tells me two things … They are trying to strike while the iron is still hot or this is all we have for the next 10/15 years :( and maybe the same centers will offer cells transplants when the time will come.
Why would they have information we don’t have….
it’s not about information, entrepreneurs sometimes sense the market, but often what they sense is wrong. We’ll see…
This is happening all over the world, demand is increasing because this subject is no longer a taboo and people talk openly about it, and also the drop in prices is already accessible to ordinary people who prepare and save for it no longer restricted to the rich and famous
Almost certainly. When we finally have some sort of hair multiplication technology it will undoutedly need hair transplant surgeons to give it effect. So it’s win win. Personally, I’ve run out of road. My hair is far too sparce now for a transplant, so I’ve decided to go the hair system road for what I hope is only ten years so so before I can have my own hair back. I can but dream. I’ve been doing that for the last thirty five years. Male pattern baldness is one of God’s little jokes that it’s difficult to laugh along with…
Hair systems are a blessing. Are they perfect…NO, but it beats just sitting home feeling sorry for myself like these past two decades. Since wearing hair, I am so much happier. My French lace system this summer is holding well and at my new job no one really knows what I looked like when i had hair loss. So happy. Cannot explain my joy since wearing hair.
Great to hear. I miss hearing from Scott, our resident Parkour and Hairpiece expert. Hope he did not get kidnapped in his travels to exotic locations.
PS does anyone know how they can be sure that the cells they are using for the hair multiplication are not themselves subject to MPB? Many men (me included) have thinned out hair in the horse shoe, and that hair can deplete further as the years go on. (my father lost his hair on top and the horse shoe hair then thinned out gradually over thirty years before he died). If you take hair cells from that area, surely there is no guarantee that they are not programmed to die off over several years. If they are the seed for cell multiplication then that would obviously be a major issue. Does anyone know if it is possible to test the cells that would be used to seed new cells to make sure they will never be subject to MPB, or is this something that is work in progress? thanks
Please share you email if there is a chance to enroll myself in the trial.
Is Dr Fukuda still holding to his 2023 plan? That forecast really looks like a random guess made five years ago and forgotten right after the interview… A bit disappointed.
Referring to the update: „in a move that COULD ONE DAY treat hair loss“
That doesn‘t sound like 2023 anymore :(
Dear admin,
Do you think having a hair transplant will impact your susceptibility to a potential future treatment like for instance the one dr. Fukuda is working on? I heard someone say the scarring of the operation might undermine one’s ability to receive future treatments.
Thanks for your research, i check in every now and then and am optimistic about all the progress!
Your today‘s update contains the paper “Reprogramming of three-dimensional microenvironments for in vitro hair follicle induction”…which was already in your June‘s update.
Thus nothing really new.
Good catch Ben. Previously, I mentioned it was not yet peer reviewed, so that must be why all the news caught up yesterday… when it was finally published. Will correct.
Good job admin! in reference to the name of the thread: “human tests in 2023”, we know that Fukuda said in 2018 that human tests would start next year… are you still hopeful on this date? Regarding the current news, it seems fascinating to me actually, to cultivate functional hair follicles artificially in vitro? who cares if they are mouse, but it is clear that this is the real cure. We hope that they will soon carry out the next step and carry it out with human cells and then carry out the clinical trial. What is your opinion??
I don’t see any reason why they wouldn’t start human trials in 2023.
I think “human trial in five years” and “human trial next year” carry very different weights. Especially if they have stop reiterating their plan when the date is closer. We all keep our fingers crossed but in a bet I’ll keep my chips against it…
While this is still very important work, I can’t help but wonder that if verteporfins success is replicated in other patients or even improved, that it will make hair multiplication a bit pointless, unless of course you have non existent donor hairs anywhere on your body
Great news. I get tired of hearing anything about mice, though. Humans. We’re humans. Forget the mice. Plenty of us humans to test on. Let’s get moving, already. Volunteers galore. We move so darn slow on this stuff.
Also, the last I heard, pigs was the way to go – not mice.
Happy things are progressing. And allow me to be the first one ever to say… A hair loss cure may be just 5 years away.
Facepalm :-)
Even if they used human stem cells it doesn’t mean that the experiment will work on human head.
This is a long process. It will take years and years of testing till we get to pre-clinical trials.
Junji published new scientific paper couple of days ago if it means something.
https://www.sciencedirect.com/science/article/abs/pii/S1389172322002572?via%3Dihub
Years and years of testing before pre-clinical?
How come Fukuda himself announced human clinical for next year already?
Given the information we have, it’s very safe to assume that „pre-clinical“ is already finished by now. Not „years and years“ „before“ „pre clinical“.
I don’t know how people come up with this wild theories, it’s crazy.
Just FYI: Tsuji was officially given green light for human trials in 2019 (!). It‘s only due to financial troubles that they had to shut down the operation and are obviously still struggling until today to find funds. Unfortunate that is.
And to add: Japan has completely different laws, it makes it much easier to conduct tests like these.
Precisely!
What I do read into Fukuda’s recent papers (microenvironment + cell sorting) is that they established a model where you can grow a hair from cells to fully sprouting – completely in a dish, fully controlled (might have been done before, but not for purely observative reasons).
With microscope/optical advancements and new AI/ML-tech they now can see the full signalling/developing/molecular pathways of a hair. Which in turn will ultimately give us full understanding of this immensely complex process and eventually will lead to a cure, and not just one probably.
It’s actually quite astonishing what they are doing and tells us that we as humans need to understand a problem as good as possible to find solutions.
Plikus („CelltoCellChat“) and Tsuji (both for hair) and Isomorphic Labs (general) have developed similar models because of that.
At least that’s what I see. Admin?
@Ben
They are doing this testings with mice stem cells. It would be totally different if they done it with human stem cells. So, from my perspective this is good sign, but still far away many years from pre-clinical trials. It is too complicated, very expensive. These kind of treatments will take decade or two till it gets to ordinary clinics.
Well then, you gotta tell that Dr. Junji Fukuda and Dr. Tatsuto Kageyama from the Yokohama National University.
They probably don’t know that!
They mentioned that in press. They need to verify their method with human stem cells.
On other hand, Stemson is working with human iPSCs on pigs. Totally different story.
Let’s be clear. This is not ground breaking paper, because mice stem cells grown much easily on mice than human stem cells. That is the tricky part.
@Bryan is on point. Bryan nailed it perfectly. He summed it up perfectly.
Kind of blows my mind we can be so close to clinical trials and it stops due to funding. The list of bald or balding billionaires (worldwide) is large (not to mention multi millionaires). Some spend 5 million on a boat over a weekend. Chump change. Heck, I watched a show with one billionaire was a pen collector – he had the pen Abraham Lincoln used (it was worth a ton). I can’t see why they don’t toss a few mill (again, chump change) to a company super close to trials in order to keep it going (and perhaps, if successful, reap the rewards both financially and, well, folically).
I am pretty sure it’s because these so-called discoveries often end up just being a lure to obtain further funding to keep departments in operation. Universities are also not exempt from this type of behavior.
I am 63 showing early signs of hair thinning ! Add my email to the trial.aka of mice & men.
What’s that?
http://www.koreabiomed.com/news/articleView.html?idxno=14642
Research paper:
https://pubs.acs.org/doi/10.1021/jacsau.2c00241
Key takeaway:
„Noninvasive, sustainable, follicle-free hair transplantation was demonstrated“
Summary:
Hair strands (NOT follicles) are being cut off, transplanted and glued subcutaneous.
That’s absurd. I mean, technically you could have endless hair transplanted. But of course it doesn’t grow, you shouldn’t cut it, and the durability is in question, and it’s still invasive (and scarring?).
Maybe to fill up a crown?
Several people emailed or posted these links recently. I was unsure of what to make of it. Reddit has some thoughts too:
https://www.reddit.com/r/tressless/comments/xkoh7t/kaist_develops_biofriendly_adhesives_for/
Ah okay. I just discovered it. Let’s see where this is heading.
But the best news by far are these:
https://amplificabio.com/amplifica-completes-11-8-million-series-a-financing-to-enable-clinical-development-of-lead-products-to-treat-androgenic-alopecia/
Probably even the best news of the year. I am very excited.
https://www.instagram.com/p/CkWArMxrOEN/ News is now diffusing into social media lol
Why is this doing the headlines, even in France? https://twitter.com/CerfiaFR/status/1586801158160539648
To me, there’s nothing to be hyped about, we dont give a to mice … what do you think admin?
Preprint of new Fukuda and Kageyama paper:
https://www.researchsquare.com/article/rs-2263496/v1
Administrator, I think that like all the users of the page you have been waiting for a clinical trial in humans of hair cloning for years (too many). I really like your optimism and the selection you make of what are the true solutions for you. I sincerely believe that Fukuda has developed the same system as Stemson and that he is willing to test it as soon as possible but… How long do you think it will take? The latest update of the mouse follicle created in vitro is just the demonstration that your system works and what else do you need? Test it with human cells in vitro? This solution is not a medicine… (and if it has shown that it is capable of creating a follicle organoid with autologous iPCS cells that is capable of producing a hair and multiplying it) with a clinical trial you will be able to see if it works or not, you don’t need do phase 1, phase 2, phase 3… phase 1000. In short, knowing that this doctor has the keys to do it, do you think he can carry it out in 2023? Let’s hope so….
Update?
There is an update on junji fukuda regarding biomaterials and 3d culture growth of hair follicles.
Bro this is incredible, if I’m understanding correctly, not only did they grow hair follicles in vitro in the lab, but they were able to get them to cycle after retransplanting them, when they move to human trials which is hopefully this or next year it will be game over for baldness, and not just AGA related baldness either ..all baldness. Because they grew hair from cells in a lab. All they need is the cells. Fukuda’s team has done what no one else has done except maybe Dr Koehler who’s team was able to grow skin with human hair on it. Amazing!
Is there any new updates from junji Fukuda? It kinda feels like junji Fukuda is most likely not going to start human trials this year like he promised .
There are some updates in feb and march 23 when you search in researchgate.com. They are actively involved. But when will they do trails is still to answer.
@gurvinder kaur Thanks bro
New interview from Fukuda:
https://top-researchers.com/?p=2510
Nothing completely new except they are pursuing 3 methods at TrichoSeeds: cell activation, cell injection and follicle transplantation (I hope I am right – the translation from Japanese is always a little wooden).
The industrial partner is Rotho Pharma.
It honestly doesn’t sound like they are ready in 2023, he says „in the next years“. Given the latest infos on Tsuji, OrganTech might be even faster then TrichoSeeds, which would be astounding to me after Tsuji‘s previous disaster.
Thanks Ben. The next few years statement seems to be in relation to method 1 (reactivation). I would not be surprised if they start human trials for at least one of these three methods by the end of the year.
Oh okay. I translated it into German (my first language).
To translate Japanese into German always gives the worst results compared to other languages – it always leaves me confused.
Anyhow, I hope you are right (I couldn’t find the part where Fukuda references the timeline to a certain approach)! A website from TrichoSeeds would be awesome though to shed some light onto their work.
Yea it seemed to be referring to method 1 but I could be wrong.
Junji Fukuda don’t have actual product. He is doing research.
On other hand, Dr. Takashi Tsuji and OrganTech have actual product and they got investment from Kobayashi Pharmaceutical to start clinical trials next year. OrganTech technology is complex and years ahead of their competitors.
Can someone explain me what are the differences between Dr. Takashi Tsuji and Dr. Junji Fukuda approaches?
Why Dr. Takashi Tsuji will start clinical trials and Dr. Junji Fukuda is still many years away?
Damn I knew it junji Fukuda is full of French fry oil. Everybody he’s not going to do nothing neither is Dr tsuji.
What a contributive comment Marc! Where would we be without you!
Oh I’m sorry yoyo I didn’t analyze the molecular compensation of junji Fukuda’s French fry oil
I wouldn’t want someone who can’t differentiate between composition and compensation to analyse anything.
This sound promising! I hope we get news soon enough about the 2023 follicle cloning hair transplantation human trials. That seems to be a definitive hair loss treatment without the need of propecia.
There‘s a new segment on Fukuda‘s lab website:
http://www.fukulab.ynu.ac.jp/Research/research_ha03-en.html
What I really like about the Fukulab is that they always have in mind to make their approach fast, cheap and effective – viable for a mass market. Their whole 3D-printer setup including all devices costs less than 50 grand (I looked it up) and is a standard printer with only a small customization (as far as I could read it from the papers). One of those could potentially serve 1000s of patients per year.
Of course there‘s additional tech needed (cell cultivation etc.) – but a future clinic could be equipped for fairly cheap money. Eventually it won‘t cost more than a standard transplant, possibly less.
Thanks Ben, very interesting! Just wrote a new post today in which I mention Fukuda.
Minor news from Fukuda. A new patent is online for a couple of weeks now.
This time it’s a technical device for transplanting the cells:
https://patents.justia.com/patent/20230210557
One of the inventors is Keichi Imai, a representative of Toppan Printing, a daughter of Toppan – another one of those Japanese giant multis (several areas of business).
It’s good to see that Tricho Seeds is looking at all angles of a possible business venture, that includes the transplant devices for clinics as well.
Nice look out, idk I thought this year we’d have at least one big news of clinical trials or something. But this year has been very slow :/
Another small update – the HFGs are (possibly) being cryopreserved during the whole process.
This should make things much easier as the manufactured follicles then can be transported anywhere in the world.
There‘s also a graphic from a tool (‚microfinger‘) which could be connected to the latest patent updates of Fukuda.
Have a look:
https://www.sciencedirect.com/science/article/abs/pii/S138917232300186X
Slowly but surely TrichoSeeds is getting there!
Sorry can you summarise how this is different to Stemson or Epibiotech, I don’t understand how this works lol
Great find Ben.
Ben Good find hell yeah slowly but surely God willing! I think if more people would pray for this it would be helpful to send out good vibes ! And junji Fukuda could bring this to fruition.
If more people would pray that wouldn’t change anything at all.
There’s also no god involved, only dedicated top researchers.
This isn’t religious.
Way to bring the mood up!
A small update:
https://www.ko-karei.com/healthcare-v/
TrichoSeeds Co., Ltd.
Hair regeneration medicine for the treatment of male pattern baldness
We provide services that treat alopecia using hair regeneration medicine. The company plans to have three pipelines for its business, and for the first one, it has already agreed to conduct clinical trials with a major domestic pharmaceutical company, accepted investment, and implemented technology transfer. The company will receive approval to conduct clinical trials by next year. Hair is regenerated by preparing transplanted tissue using unique cell culture technology and transplanting it to the skin where hair loss has occurred.
Admin good find but what does that mean exactly is junji Fukuda/trichoseeds going to start human trials for hair cloning next year or a topical treatment because I know there working on a topical treatment and a cell injection and actual hair cloning, I guess what I’m asking is which of there treatments will go into human trials by the end of 2024 ? Admin would be very helpful if you or someone can explain this to me where a simpleton like me can understand it thanks.
Another remarkable paper dropped from Fukuda:
https://www.nature.com/articles/s41598-023-40521-x
Very interesting, please read the part „Discussion“ for a quick summary if you don’t want to get through the whole paper (although recommended!).
Seems they are optimistic that OTC could be good enough as a hair growth agent.
What stuck out for me the most though is that they are already using their own invention („Follocloids“, engineered hair by their own method) in a testing environment for possible hair growth agents. Which proves they cracked the code of „cloning“! Of course there‘s other factors in play, but the core tech seems to be working. Great news imho.
The trials next year should be interesting for sure. Good find Ben! Make sure Marc doesn’t get anywhere near this he’ll add 10 years to it
New segment on Fukuda‘s website…
http://www.fukulab.ynu.ac.jp/Research/research_ha04-en.html
Hi Ben,
FYI — A few months ago I added the below link in the post (end of third paragraph) when it was new.
http://www.fukulab.ynu.ac.jp/Research/research_hair-en.html
Oh yes I know that, but thanks for pointing out.
I‘m just saying that there is a new segment on that sub-page called „Transplantation of hair follicles generated in in vitro organoid culture“, here: http://www.fukulab.ynu.ac.jp/Research/research_ha04-en.html
„The transplanted hair follicles stably remained in the skin and repeated hair cycles for at least 5 months. Notably, hair shaft orientation was controllable by the direction at transplantation. These results demonstrated our approach is promising for highly oriented hair regeneration.“
Which is crucial for an aesthetic outcome.
Hello administrator, as we approach the new year, I wonder when there will be any update from Junji Fukuda about his clinical trials. Happy New Year to everyone!
https://www.youtube.com/watch?v=c8dGm_LKH5Y
In case no one has seen this video. You should translate it to English first using “CC”.
Can you summarize the content of the video a little for those of us who don’t speak English Yoyo? Thanks in advance!
It wasn’t saying anything new in particular, except that they believe that this will be able to come to fruition in the very near future, and how their technology has improved to now make nearly 100% efficient hair follicles.
Hey Yoyo – just to remember, the video is already almost 2 years old.
I hope they announce their trials soon, it’s time.
Yup you’re right Ben, on the other hair loss blog they did say they are wanting to start clinical trials this year. But what are your thoughts on Amplifica? We’re supposed to see results Q1 2024. I like that they’re using “natural” components it could help push the product quicker to the market.
I‘ve become very cautious with predictions, it‘s always much more likely a candidate fails intead of succeeding.
But Amplifica is in my Top 3 along with HopeMed and Fukuda. Plikus is an absolute mastermind (watch his latest Uni-talk on YouTube!), and I think the trialled candidate is quicker at the market as it is used already in other conditions.
Btw. have you see Stemson‘s new method to anchor the hair-cells? They scrapped their „lolli-up“ and now use a thread – exactly as Fukuda does. Makes me wonder how much time Stemson lost with the wrong approach and how far Fukuda is ahead in the game.
Wait I didn’t hear about Stemson using thread, have you got a link? Their lolli-up method theoretically seemed to be a great idea for transplantation. I kind of lost the excitement because of the trial expected to start in 2026, assuming they are wanting to do it in US, if it’s in Japan, S Korea or Australia I’ll be back on the train lol. But yea Fukuda method and timeline is something to be excited for, the only thing is safety for me lol. Regarding Plikus I definitely agree he’s intelligent I just hope the results won’t be as disappointing as Kintor’s (not that I had high hopes to begin with) but I do have a feeling Amplifica and Pelage will give us some good results for robust hair growth. It’s plausible for Amplifica’s candidate to come to market around 2026.
Thanks Yoyo!
No worries Alvaro.
New Fukuda-paper:
https://pubs.acs.org/doi/10.1021/acsbiomaterials.3c01346
Seems like a continuation and improvement of previous approaches – they are constantly developing and tweaking.
The video is quite interesting: with this speed you could easily produce a couple of thousand droplets/follicles within an hour with only one printer. Which is essential for cost-efficiency.
Thanks Ben! Too bad its in mice, but still very promising.
“Using human hair follicle-derived epithelial and mesenchymal cells expanded using these approaches, the optimization of the conditions for preparing mfHFGs will be the focus of future studies.”
I think those „future studies“ can only be human trials. Fukuda also mentioned somewhen that human cells are being used too.
As far as I understand this paper is mainly to demonstrate the functionality of the devices.
Fukuda did also mention that he was to initiate clinical trials this year
Minor news. Small report plus video regarding the Oxytocin-study.
https://www3.nhk.or.jp/news/html/20231229/k10014302541000.html
I now understand why they tried Oxytocin of everything – apparently pregnant/breastfeeding women produce significantly higher amounts of Oxytocin and this could be the cause for thicker hair (a known phenomenon) in those women.
The live-model of Fukuda‘s drug-testing for hair could become the real revolution along the way.
@YoYo
Sorry I forgot to reply to your question. If you watch the video from Stemson…
https://youtu.be/xCj7lBICpkw?si=V_mTAdvR3Mo5t1k1
…at 06:10 you see in the third window of that slide that they now use a „guide“ which looks very similar to Fukuda‘s thread. In the next slide (08:24) this guide is implanted into to skin of the mouse.
I think it’s a good sign if both ventures follow the same approach, as it must certainly work with that method.
I disagree with you in terms of safety – both are autologous and that’s supposed to be safe. IPSCs is probably different and poses a bigger risk.
Hey Ben no worries! I’m a little confused because that “lolli-up” slide was previously used to “mix the iEP and iDP cells” and then guided via a scaffold onto the scalp (Unless I’m understanding it wrong). And for clarification are you saying that both Stemson and Fukuda should be safe for treatment due to their autologous behaviour? Thanks!
Fukuda, Tsuji, Stemson, Hairclone, Epibiotech are all autologous.
Stemson and Epibiotech are also pursuing allogenic solutions ( ipScs), but these are definitely further away. Kangstem I am not 100 % sure if they are autologous or allogenic, based on the information available it autologous too.
I cannot explain detailled differences of these contenders from a technological or biological viewpoint. It feels though that they became more and more similar over the years.
This research makes all the sense in the world Ben, in my case I started losing my hair when I was 19 due to the stress of studying at the university, the main hormone that is secreted by stress is cortisol and at that time I began to having a lot of itching on the scalp due to excessive secretion of sebum, in which there is a high concentration of DHT, which in a matter of weeks caused my hair to thin completely and fall out abundantly, triggering androgenetic alopecia. Therefore, a medication based on oxytocin (as opposed to cortisol) could have the opposite effect, although this is demonstrable since when we are stressed our hair falls out and when we are happy and calm our hair becomes stronger. But for those of us who have androgenetic alopecia in our genetics, only cloning is the definitive solution.
Follow-up on their Oxytocin-study:
https://www.researchgate.net/publication/378520246_Cinnamic_acid_promotes_elongation_of_hair_peg-like_sprouting_in_hair_follicle_organoids_via_oxytocin_receptor_activation
They again used their own new proprietary method „follicloids“ to validate the effects of Oxytocin on DP-cells. It’s quite fascinating and this tech will most certainly lead to a much quicker R&D for new compounds against hair loss.
The results itself are also remarkable imho. This might be a new and interesting avenue.
I bookmarked the Nature article on that paper recently and was thinking of writing a post. But I have two others on the backburner too!
https://www.nature.com/articles/s41598-024-55377-y
What about this new research from the lab? https://phys.org/news/2024-03-cinnamic-acid-developmental-avenue-hair.amp
Remember: mankind has probably been working on the “hair loss problem” for as long as humanity has been on Earth. Millions of years? Billions of years? That’s fine. Other health-related concerns have been solved, and this is just one more challenge!
I have had many hair transplanting sessions done. Were they worth it? There is also the cost of taking Propecia daily. Was that worth it? YES.
So, someone needs to start thinking
WAY outside-of-the-box. It’s not just a corny phrase. :D Or, one day one of the smartest people to ever exist will solve the problem. As long as other people are not harmed by the research attempts, I believe that the hair problem can be solved.