Follicum
I am on Follicum’s mailing list and the company had four significant developments during the past month that were deemed worthy of e-mailing to subscribers:
- On January 18th, Follicum announced the identification of key receptors in human hair follicle cells to which the company’s lead hair loss candidate (FOL-005) binds. FOL-005 is a modified version of the endogenous protein, osteopontin.
- On February 2nd, Follicum announced the launch of its English language website. It is worth going through the menu titled “Hair Growth”. The Swedish version of the site remains online.
- On February 6th, Follicum CEO Jan Alenfall gave an interesting interview to Aktiespararna. He discusses both the hair loss product as well as the new diabetes product FOL-014.
- Of most important to us, on February 7th, Follicum announced that it had received go-ahead approval from the German Medicines Agency (BfArM) and German Ethics Committee. This is to commence a Phase IIa clinical trial in Germany in relation to FOL-005 on human patients. The company will partner with Clinical Research Center for Hair and Skin Science (CRC) in Berlin and bioskin in Hamburg.
Follica
In 2016, Follica announced that it was going to address its hair loss treatment via the acronym “RAIN”. At the time, I guessed the “R” to mean Regeneration, and the “N” to mean Neogenesis. Earlier this month, blog reader “PinotQ” notified us that Follica owner Puretech most likely recently updated its website and now spells out that the “AIN” stands for Abrasion Induced Neogenesis. Perhaps we just missed this development last year, but it is important enough to spell out here.
French Fries
A few days after I covered the groundbreaking work of Dr. Junji Fukuda, major newspapers around the world figured out a different (i.e., clickbait) take on the story. One that clearly got far more publicity and Facebook likes. They labeled Dr. Fukuda’s discovery as “Chemical in McDonald’s French Fries Could Cure Hair Loss” plus other minor variations of that title.
All because the chemical (dimethylpolysiloxane) used in the McDonald’s french fries via the oil fryers was involved in part of Dr. Fukuda’s research, even though it had no direct influence on hair regrowth. This french fries fable has since became the biggest hair loss related story of the year, and is unlikely to be surpassed in superficial importance for the rest of this year.
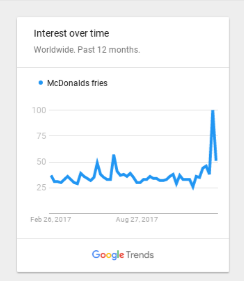
Unbelievably, numerous blog readers who already read my original post on this research still thought that the McDonald’s fries story was something totally different and perhaps worth looking into.
I had to delete the repetitive reader comments about this subject in recent posts and did not bother to respond to any e-mails abut this story.
The best advice comes from Dr. Fukuda himself:
“I have seen online comments asking, ‘how many fries would I have to eat to grow my hair?’” he said. “I’d feel bad if people think eating something would do that!”
Other Items of Interest
— Some new companies and increasing competition in the South Korean over-the-counter hair loss treatment market.
— Replicel gets new investment from Chinese company YOFOTO. Seems like the hair loss product related treatment rights were not granted, probably due to Replicel’s binding agreements with Shiseido regarding the Asia region?
— Since JAK inhibitors have been working on many alopecia areata and vitiligo patients, I have followed both conditions more closely in recent years. Here is an interesting alternative story about a vitiligo patient.
https://t.co/mr0EfFFDvP
Samumed is joke! Replicel not until 2023. Follicum maybe worth watching until 2020.
Follica is slumber sleep and brotzu is joke.
Let’s not fight for jak because it’s going to release phase 2 results next year not to market.
chill bud.
They keep postponing. It s quite clear for some reasons these products are not wanted. There are bigger interests for some companies atm. Quite lame. I also keep seeing those lame news that they found a new cure, one week later a new cure and so on. Each week/month a new cure. Nobody cares as long as I don t get any benefits. If that new cure isn t available I really don t care. By the time the most recent “cures” are available I ll retire prob not even my grandchildren will see it. Pathethic.
I am curious to see a photo of Follica’s results on a human head ! If i read it right on their website, it says 3 to 4 times more effective then current treatments ! That should be a significant gain when used with fin or dut. I’m guessing there probably is some form of minox among other drugs in the topical part of that treatment .
If you compare with Rogaine’s pictures on their results section, 3-4x that would totally fill in the bald spot those guys have.
Apply those same results to the hairline etc, and that is definitely a functional cure. Hell, studies like Dhurat are already superior to conventional treatments.
Someone on HLT just shared another study that found wound neogenesis even works (albeit not nearly as well) with no additional compounds in geriatric patients!
I have little doubt that this and stem cell cultivation will become “the cure” in the coming years.
That study was discussed here fyi:
https://www.hairlosscure2020.com/brief-items-of-interest-january-2018/
Interesting that CarthroniX wants to stimulate the JAK/STAT pathway in order to regrow hair, while Aclaris wants to inhibit it.
Wow I missed that completely! Strange if both ways work.
I’m no expert but this is probably a very good indicator of how complex this all is. After taking a little deeper dive into the research on RCGD 423, I noticed that the initial articles talk about the JAK/STAT pathway “The first drug, called RCGD423, activates a cellular signaling pathway called JAK-Stat, which transmits information from outside the cell to the nucleus of the cell.” However, the latest articles all talk about the Gp130 receptor: “This molecule, called the glycoprotein 130 (Gp130) receptor, receives two very different types of signals: those that promote cartilage development in the embryo, and those that trigger chronic inflammation in the adult. RCGD 423 amplifies the Gp130 receptor’s ability to receive the developmental signals that can stimulate cartilage regeneration, while blocking the inflammatory signals that can lead to cartilage degeneration over the long term.” My amateurish assumption, piecing the 2 bits of info together is that, the JAK/STAT pathway is just that, a pathway, which presumably in this case, allows the RCGD 423 molecule to “transmit” information inside the cell to the Gp130 receptor.
A couple of other quick comparisons to the Aclaris findings: Both shut down inflammation, ……..perhaps soft JAK’s do so by preventing some inflammatory molecule or trigger from entering the cell. One of the articles on RCGD 423 states that “For the drug to work, the molecules must be injected directly into the joint, where it can start to repair the damage.”. This would be consistent with the Aclaris findings that for AGA, soft JAK’s need to be applied topically or at the source of the problem……..maybe they too activate stems cells inside the follicle.
Hey admin,
I was curious about Follica’s treatment. Could it have the potential to revive follicles that have been dormant for some time or just shrunk down to small blond hairs?
The reason I ask him sometimes in certain lights, I still have my frontal hairline although the hairs are just peach fuzz.
Their goal is to create brand new follicles. I do believe their topical will revive miniature hairs but I could be wrong. They are hush hush lol
Hey Guys,
what I just wanted to ask is whether someone could briefly describe the current state of Follica and Replicel. I was out for few months
Best
Its like nothing happened bro…
Admin,
https://www.ingentaconnect.com/content/asp/jbte/2018/00000008/00000001/art00011
Does anyone know what the estimate price will be for the follica treatment?
It would be nice if sometime soon Getting All Our Hair Back Was As Easy As Getting McDonald Fries, and cost about the same.
Just not trying to take all this too seriously. Life. goes on with or without hair.
Nasa_rs
Ah… alliteration. Clever title Mr. Admin.
It was “slick” of me:-)
lol
If I was to enter this market, I would do blood tests on native American tribes immune to hair loss. No other ethnic groups in the world have this trait. Asians generally to a lesser extent, which is why hair transplants in Turkey and India are so easy, cheap and successful, but nobody else completely immune.
Just proof that the cure is already present, unlike cancer that affects every ethnic group and every species of animal and plant; we just can’t copy the cure yet.
I remember reading about a western scientists who went to an island where no-one went bald, and he found that they were all deficient in 5 alpha reductase or something to that effect.
Honestly, if all human males reduced DHT by close to 100 percent starting in their teenage years via medication, very few people would ever go bald before age of 40-50. Thereafter, other aging induced factors besides DHT could cause hair loss in more people.
But who wants to take such drastic preemptive measures at such a young age?
Stem Cells and JAKs:
Maybe the combination of injected Stem Cells supported by JAK Inhibitors is the answer?
http://www.bloodjournal.org/content/early/2018/01/23/blood-2017-06-792614?sso-checked=true
Another Stem Cell Company. (I have Emailed them and asked if this is maybe possible to treat Hair Loss)
https://renovacareinc.com/
Reenovacare answered:
Thank you for your message to RenovaCare. Out technologies have so far only been used to treat acute, severe partial-thickness burns. We are researching the best ways to utilize the SkinGun™ in treating a wide variety of skin injuries – chronic wounds, diabetic ulcers, surgical wounds, pigmentation disorders, scar repair, and other aesthetic and reconstructive therapies.
The SkinGun™ has been used experimentally on over seventy burn patients in the United States and Germany but is not currently available for clinical use. At this time we are not conducting clinical trials.
Did anyone see that they are now offering topical dutasteride?
Yup, but its in Belgium and I am not sure which pharmacy makes the product for Dr. Lupanzula, nor whether it can be shipped to other countries. Worth following this story, but I am not too optimistic after all the issues Dr. Hassson has had with trying to sell his topical Finasteride in the US. Dutasteride may be even harder to sell as a hair loss treatment in the US or EU.
Well if they sell it in Belgium you can ship it to all EU countries. Which Company is it? Are there before/after pics?
https://www.baldtruthtalk.com/threads/28669-Topical-Dutasteride-Available-From-Dr.-Lupanzula-and-Medikemos-Hair-Restoration?p=257886#post257886
Dutasteride is not approved as a hair loss treatment in the US or EU. I wonder if they can still ship it throughout the EU via using some kind of online prescription or personal doctor prescription for hair loss treatment purposes?
You obviously cannot say that topical Dutasteride will be used for prostate enlargement treatment (as you can with oral)!
A hair transplant clinic in Brussels with an UK tel. number without transparent location info selling unapproved Drugs …. Nice! :-)
Approved for BPH (enlarged prostate) treatment everywhere. Approved for hair loss treatment in Japan and South Korea.
There is the possibilty of a legal off label use of medicinal in europe, although not many physician will get into..as you get all the responsabilities.
In my opionion it would be as effective if not more if you do your own topical, after some research how topicals are created, its not that difficult.
Yup. Just this last week I got a prescription from a Canadian doctor for topical Finasteride. But the pharmacy wouldn’t fill it and ship it to me in Mexico because I’m a US citizen. US imperialism and drug companies know no bounds.
Thanks Phil. Have not done much research on Follics FR12. Have you tried it or heard from others who have?
Admin, my plan was to purchase Follics FR12 from minoxidilexpress.com since they’ll be selling it soon. But I can’t buy anything from them due to PayPal (xoom).
I’m now trying to find another way to purchase Follics fr12, since it can’t be sold within the United States….despite it being manufactured in the US!
Brotzu lotion update fyi:
http://www.unionesarda.it/articolo/salute/2018/02/15/lozione_brotzu_la_nuova_cura_per_combattere_l_alopecia-69-697901.html
Translation?
KEY takeaways–they need to stabilize it to be able to ship and actually use… if they can’t, then they likely won’t release it.
The Unione Sarda.it ” Health ” “Brotzu Lotion”, the new care to fight alopecia
Health
“Brotzu Lotion”, the new care to fight alopecia
Thursday 15 February at 10:10 – last updated at 10:41
will perhaps from Sardinia in the not too distant future treatment against alopecia and baldness. The hopes of thousands of Italians affected by physiological or pathological absence of hair were put back in “Brotzu Lotion”, prepared created a few years ago by a team of researchers from Cagliari guided by Giovanni and Giuseppe Brotzu, respectively son and grandson of the great physician and pharmacologist who in 1945 contributed to the discovery of cephalosporins, the first class of antibiotics that in subsequent decades changed throughout the world the therapeutic approach toward diseases at the time very serious.
A long road – a worldwide revolution that his descendants closer can now bisse precisely thanks to the miraculous lotion, at least judging from the results of the preliminary test. But to curb the enthusiasm of many, exploded out of all proportion with the word of mouth, we thought the same Brotzu, John understood. “The road toward the marketing of the product has not yet been completed – says the surgeon vascular ottantatreenne today – in these years we have given in the patent for a pharmaceutical company padovana with which we are continuing to work to stabilize the prepared and make it as suitable for distribution to the public”.
Many requests – a slowness then motivated, but still heartbreaking for those who expect from a life of rediscovering the hair lost. So much that they have led most of a skeptic to cry out to the Plot to Silence apparently unjustified fell on the discovery.
“I have the mail clogged by who asks me news on the lotion and the phone does not stop ringing, but there is no mystery – ensures the doctor – we are at the final words and soon will understand if the treatment will have the requirements of reliability to enter into trade”.
While we have thought the results of clinical trials, conducted years ago during the initial stages of research, to strengthen the hopes of patients. “The lotion works – ensures without hesitation Brotzu – was tested on 60 volunteers and has confirmed its effectiveness”.
On WHO ACTS THE PREPARED – The beneficial effects of the preparation were evident in subjects suffering from various forms of alopecia: from that androgenetica (better known as baldness) than areata, aesthetically unpleasant for track of absences of hair circumscribed patches on the scalp.
The intuition – yet the dream of a world populated by flowing foliage is never jumped in head to Brotzu before some year ago. The origins of the discovery potentially historic lurk in fact behind a brilliant intuition.
“We started our studies to find a solution to some of the side effects of diabetes – reveals the surgeon – specifically to defects of the microcirculation that cause annoying and painful ulcers. The same that the broad lines also trigger the degeneration of peliferi bulbs”. The therapy does not a case, not only healed injuries, but it was also grow abundant hair on the treated area. A result that has been made to turn on a lamp in the head of researchers so as to give life to new research in a field that awakens notoriously from time the attention of those who have lost their hair.
The test – “Testammo for the first time the lotion on a girl and a nurse with which collaboravamo – Brotzu recalls -, with excellent results. In the course of the last few years we have patented the lotion and sold the rights to the pharmaceutical company Fidia to complete their studies. A decision taken with the awareness that would be served a structure suitable for the final trial. I ultimately are a surgeon and now I have a certain age that allows me to leave the bulk of the work to others”.
Not even google translates that
If that doesn’t come out because of stability issues then I’m sorry but fidia has a bunch of clown scientists working there.
Quiescent Tissue Stem Cells Evade Immune Surveillance.
Agudo J1, Park ES2, Rose SA2, Alibo E1, Sweeney R2, Dhainaut M1, Kobayashi KS3, Sachidanandam R4, Baccarini A2, Merad M5, Brown BD6.
Author information
Abstract
Stem cells are critical for the maintenance of many tissues, but whether their integrity is maintained in the face of immunity is unclear. Here we found that cycling epithelial stem cells, including Lgr5+ intestinal stem cells, as well as ovary and mammary stem cells, were eliminated by activated T cells, but quiescent stem cells in the hair follicle and muscle were resistant to T cell killing. Immune evasion was an intrinsic property of the quiescent stem cells resulting from systemic downregulation of the antigen presentation machinery, including MHC class I and TAP proteins, and is mediated by the transactivator NLRC5. This process was reversed upon stem cell entry into the cell cycle. These studies identify a link between stem cell quiescence, antigen presentation, and immune evasion. As cancer-initiating cells can derive from stem cells, these findings may help explain how the earliest cancer cells evade immune surveillance.
I really wish i was smart enough to understand the info you post. It’s probably important stuff.
Patent by sangamo theraputics using crispr to cure aga
https://patents.google.com/patent/WO2017197141A2/en?q=Androgenetic&num=100&oq=Androgenetic&sort=new
Thats wild. Sangamo is a pretty serious zinc finger gene editting company.
Interesting! I remember back in 2001 they said hair cloning would be out in 10 years at max, then gene therapy would be the ultimate cure but it would be 20 years away. Maybe crspr is the only real cure. I wouldn’t be shocked if it was easier to cure aga via gene therapy than cloning….
Thanks very interesting and I might write a post on it in the near future.
Too bad the company does not mention AGA in its pipeline or trials:
https://www.sangamo.com/product-pipeline
https://www.sangamo.com/clinical-trials
Gene edditing is a sure fire check mate. I dont believe this will be available for the next two decades maybe in China but not here.
Did you browse their site at all?
There are already phase I/II clinical trials being conducted on monogenic diseases.
If successful, these treatments could be available within the next five years.
zinc finger genetic editting androgenic alopecia in living adults out at market in the next five years? you sure?
I live in Hamburg and think of participating in the trials.
Pros/Cons?
That would be very interesting that not only Brotzu, Follicum tries to do research on both diabetes and hair loss.
Activation of the Wnt/β-catenin signaling pathway plays an important role in hair follicle morphogenesis and hair growth. Recently, low-level laser therapy (LLLT) was evaluated for stimulating hair growth in numerous clinical studies, in which 655-nm red light was found to be most effective and practical for stimulating hair growth.
Not that it means anything in particular, but Puretech seems to be active in updating it’s Follica pipeline page. As you know, last week I noticed the descriptive update “Abrasion Induced Neogenesis”. Yesterday I was checking some info in the links and noticed that under this new description at the top left, they had added (RAIN). Interestingly, today I now see they have changed the description to “Skin Abrasion Induced Neogenesis” but still with “(RAIN)” underneath. I can’t be sure but the first paragraph also appears to have been modified to emphasize the focus on abrasion: “Our technology is the first, to our knowledge, designed to create new follicles and hair through mild abrasion of the skin, followed by treatment to enhance the effect.”
http://puretechhealth.com/our-pipeline/wound-induced-neogenesis-rain The Follica website does not appear to have changed as far as I can tell.
Also, I hadn’t really thought it thru before but, if you recall, Puretech had updated the expected timing bullet on their website to disclose 2 studies beginning in 2018: “Our regenerative biology program is currently progressing into a pre-pivotal pilot optimization study expected to start in early 2018, with a pivotal trial expected to begin in the first half of 2018”. It got me curious as to exactly what is a “Pivotal Study”. Wikipedia describes it as : “A pivotal trial is a clinical trial or study intended to provide evidence for a drug marketing approval, e.g. by the United States Food and Drug Administration. Phase III trials are assumed to be pivotal so the phrase is often used for the rare pivotal phase II trials.” So apparently, this is that rare instance where Pure Tech doesn’t intend or feel they need a Phase III trial.
Very interesting if true.
I will say it once again Follica is the only thing coming out before 2020 for us in the USA. I’m sure they have already collaborated with derms and ht offices to distribute the RAIN dermabrasion procedure. The big question is how well with this work? We talking some new hairs here and there with thickening of existing hair or we talking massive regrowth 100cm2 hairs with nw5 to nw2 growth. If they can even increase 20cm2 with legit terminal hairs and stop further loss then this is a true game changer.
Pure speculation on my part (see what I did there? :) ) but my interpretation was they were initiating Phase III the first half of 2018. In part, as their pipeline chart shows the arrow 1/2 through Phase II, but who knows.
Either way, this appears to be their go for approval study. I also noticed that under “Indications”, they have “Epithelial Aging” under “Androgenetic Alopecia”. Not sure if that is new either but it might explain the recent addition of ‘Skin” before “Abrasion Induced Neogenesis”. The Cotsarelis research had always indicated that after wounding the repair process could be tweaked one way or the other to make hair or skin. They appear to be suggesting that they may know enough now to influence the repair process either way.
They will delay anyway. They have planned to start this pivotal trial for several years now. They will postpone again.
They have delayed their pivotal trial several times, however, we are a third of the way thru the first half of 2018, Puretech is actively fine tuning their Follica pipeline page; added a second “pre-pivotal” trial reference; and left unchanged the 1st half of 2018 timeline for pivotal trial initiation. Those are all reasonably good signs that they are or will be moving forward in close approximation to their timeline. I think it is also a good bet that if this does in fact turn out to be the “pivotal” trial they go to the FDA with, they are probably using compound(s) that have already been FDA approved.
So when do you expect follica to hit the market? (best case scenario)
I have tried to avoid projecting timelines because they are really pointless until someone shows incontrovertible proof that their treatment consistently works at an undeniably higher level than anything available now. Until that time comes we are really just guessing based on our level of optimism/pessimism about something we really know little about. There is a lot going on and research is knocking on the door from many angles but we still having nothing concrete to show for it. But if indeed, this is a go to the FDA for approval Pivotal trial (which no one with a game changer has done as far as I know), assuming the pivotal trial has respectable results, (which their confidence in seeking approval without a full phase III may indicate), and if this is based upon the use of a compound already approved by the FDA, my guess would be 2020. I don’t think they would come out with a treatment if it wasn’t better………..the real question being how much better. Keep in mind, we haven’t seen any evidence to date that it really works………..possibly because it doesn’t work that well or because it is easily duplicated.
Will someone break down follicas release date
Let me open up a can of worms here.
Has anyone ever researched, or even experienced, positive or negative effects for hair loss/growth in relation to the use of narcotics; in particular cannabis?
I ask purely from a scientific standpoint. Please don’t reply with any moral objections.
Tommy Chong is balding and Cheech has been bald for years. Isn’t that all we need to know?
I think that alcoholics seem to have better hair than non-alcoholics. Possible reasons could be higher estrogen and lower testosterone from all the booze over many years.
However, one time we discussed this on this blog, several others did not agree at all.
Of course if someone becomes an alcoholic after he has lost most of his hair, the regrowth will not come.
Alcohol also deprives the body of nutrients and dehydrates. Alcohol inhibits antidiuretic hormone (vasopressin) from the posterior pituitary gland, causing increased urination. Also blunts cognitive functioning.
Ok we all know alcohol is bad for you beyond minimal quantities. Are you trying to implying something about how alcohol benefits hair via damaging everything else? Or was that just a warning to not drink?
In moderation, particularly red wine is best since it contains resveratrol.
Or maybe the alcohol keeps them away from stress..lol
Or they are already stressed, hence they turn to alcohol.
@Mjones: if Follica can really advance few NWs then it will definitely be a cure in combination with HT. say u r a NW5 and then it will make u NW2 and with top notch surgeons they can make u NW0 in no time… still I dont believe Follica possesses this improvement
I hope they can donitello. I REALLY hope they can. Or at least their claim of being 4x better than current treatments be real and that it works for everyone. I remember follica saying that their RAIN treatment would be a hair transplant replacement. So that is a good sign for a lot of regrowth. If RAIN can grow 20cm2 terminal new created follicles, then thicken up current dying hair that could really produce excellent regrowth. I think if you are a nw4 or lower this could potentially be a cure. Follica gets you down to a nw2 then get top notch ht from rahal or bisanga and you are nw1 or thick nw2. Let’s hope for the best and that we have a potential new treatment by the end of this year. It will be exactly 20 years since the last approved hair loss treatment Propecia. 1998. We should have had at least 3 better treatments by now.
It really is a make-or-break year for some of the big players. Interesting times. There will be less players to read up on by the end of this year whatever happens.
My money is on Shiseido. I think the rest are just a bunch of have-a-go chemists.
@Scott: Shiseido? Really? I don’t think so.
GREAT BLOG.
Follica, Shiseido and Tsuji are the only ones with tech worth getting excited about.
Everything else has either:
A) Already failed
B) Mouse experiment that might begin human trials (maybe) in 15 years.
Wrong. Aclaris has started human trials.
They’re in the “already failed” category
‘Fevi, Brotzu* Supposively they are presenting results on brotzu on the 15th of April. Who knows if it’s true
What about cannabis strains with high CBD and low THC?
I heard allot good things about cbd.
This is the discussion I tried to put forward but everyone instead just spoke about alcohol :-)
Hey Sam I ve been doing rick Simpson hemp oil for two years now,I take a grain size before bed and sometimes in the morning. Even tried it topically,with no real growth, but for other things like weight loss was unbelievable,maybe because I slept the whole night and not getting up every hour. Everything set back to like I was twenty years old again. Except for my hair. Always figured people with ed from fin should consider trying the oil and it may counter act it. It was like it reset everything . I still take it now , cancer runs in our family and this oil is know to be a cancer cell killer.
I recall reading that there is a company that pure tech or Penn or Cots hired to work on creating a topical based off the research that has been done the last few years. Does anybody recall the name of the company and/or know how they are progressing?
@Tom
You talk about Kythera ? PGD2
https://www.hairlosscure2020.com/category/kythera-biopharmaceuticals/
Every week a new theory …
http://onlinelibrary.wiley.com/doi/10.1111/jocd.12501/abstract;jsessionid=E9D4BC4358B266FBABD5A0F8A75B109A.f03t03
Interesting how it works for BOTH AA and AGA hairloss. Just like MY Theory that JAK works for Both AA and AGA. Just that the AGA needs a different delivery method to get into specific skin layers.
@Ryu…nope
What about Follics?
Why can’t I buy this stuff in the United States? Acne prescription medications have gone over the counter (adapelene gel). Why can’t topical finasteride be purchased over-the-counter in the United States?
Follicum will have the best product, it´s the most serious company of the ones listed at this page. It will not take much more time before a larger company buy the rights in Fol-005.