The cost of generic dutasteride is drastically cheaper than the cost of brand name dutastride (Avodart).

Update: January 9, 2022 — Yesterday, for the first time ever, I purchased my generic dutasteride 0.5 mg using GoodRx. Please note that you can sort on their site by number of pills as well as branded versus generic. My cost for 90 pills was just $23.31 via Safeway (my closest pharmacy). This is a lot less than what one would pay with regular health insurance coverage (see table at bottom — not updated recently). I get my prescription from Medical Wellness Center.
In the past, I used to go to Costco, but I ended my membership there last year. GoodRx will save you 95 percent of the cost of your pills (or capsules) if you do not have any health insurance. The product I purchased was made in China (see image on right) via Epic Pharma and its local partner Humanwell PuraCap Pharmaceutical. In the past, I have purchased my generic dutasteride from various other company names, including Breckenridge, Apotex and Camber.
Generic Dutasteride Price
After I wrote a post on the cost of generic finasteride last year, I received several requests to write one on the cost of generic dutasteride. However, the reason for my not being able to do that right away was because the patent for Avodart (the original brand name dutasteride that is made by GlaxoSmithKline) only ran out late last year. It took a few more months before generic Avodart became readily available.
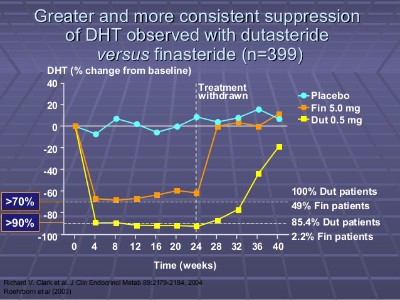
Note that dutasteride is only approved to treat hair loss in South Korea and Japan as of the time of writing this post. In the US and EU, it is approved to treat enlarged prostates, but not yet for hair loss. I am optimistic that this situation will change in the next several years, but no guarantees. Edit: No longer optimistic.
As I have mentioned many times on this blog before, dutasteride (see positive and negative reviews and testimonials) is likely to give substantially superior results in comparison to finasteride when it comes to treating hair loss. But at the same time, likely to cause more frequent side effects.
Do your research carefully before deciding to proceed with such a strong drug and always discuss taking it with your doctor before proceeding. In rare cases, it seems like the side effects from both finasteride and dutasteride can be permanent.
GoodRx Discounts
Note that various online sites now offer coupons for drug purchases if you are buying them with cash and not paying via insurance. Perhaps the most popular one is GoodRx, which has coupons for numerous drugs, including for dutasteride. For those not in the US, feel free to provide your country’s prices and discount options in the comments to this post.
Note: Prices and brands at the same pharmacy chains may vary by US state. Also keep in mind that many stores may price match or have internal savings plans.
Generic Dutasteride Cost Table
Country of manufacture entries in the below table could be off and I have not tried to go through product brochure attachments on company websites for now to try to find out.
Teva Pharmaceuticals is headquartered in Israel, but a pharmacist told me that their generic dutasteride was made in the US. The Breckenridge Pharmaceuticals website says it is based in the US, but a Costco employee told me that the packaging on their generic dutasteride says its made in New Zealand.
Amneal Pharmaceuticals website says that 95 percent of its products are made in the US and 5 percent are made in India. GSK is headquartered in the UK, but it seems like the company’s Avodart is made in the US. Please let me know country of manufacture if you purchase any of the below and find errors in my table.
| Pharmacy Name | Avodart (0.5 mg) — 30 pills | Company Name (Country of Manufacture) | Generic Dutasteride (0.5 mg) — 30 pills | Company Name (Country of Manufacture) |
| Costco | $190 | GSK (US) | $21 | Breckenridge Pharmaceuticals (New Zealand) |
| CVS (Target now seems to house CVS) | $207 | GSK (US) | $172 | Teva Pharmaceuticals (US/Israel) |
| Rite Aid | $222 | GSK (US) | $173 | Amneal Pharmaceuticals (US) |
| Safeway | $232 | GSK (US) | $203 | Amneal Pharmaceuticals (US) |
| Sam’s Club | $190 | GSK (US) | $100 | Teva Pharmaceuticals (US/Israel) |
| Walgreens | $206 | GSK (US) | $182 | Amneal Pharmaceuticals (US) |
| Walmart | $195 | GSK (US) | $157 | Teva Pharmaceuticals (US/Israel) |