April 2, 2023
An encouraging update on Verteporfin and some donor area hair regrowth from Jordan-based Dr. Barghouthi in his interview with the Hair Transplant Network. Still too early to know whether these results are permanent and constantly replicable by other surgeons. And whether any new hair will cycle ok in the long term. But this initiative is really great to see.
Please read my entire original post below for the relevant studies and past videos. It is also possible that FAK inhibitors will be even better than Verteporfin in regrowing hair based on concurrent research findings at the Longaker Lab.
July 24, 2022
It is Verteporfin week in the world of hair loss. More than a year after Stanford’s Longaker Lab findings that Verteporfin injections cause regenerative wound and skin healing in mice. Back then, the news even made it into the NY Times: “Imagine, Surgery Without a Scar.”
Besides the three new recent Verteporfin and hair growth related videos that I mention at the bottom of this post, this week I also got: Verteporfin related emails, blog comments and even a private message on Instagram. I am not as excited as so many people when it comes to the hair regeneration prospects, but this subject is clearly going to remain important in 2022.
Do not Forget FAK Inhibitors
As I mentioned last year in a post, FAK inhibitors might be superior to Verteporfin when it comes to wound healing and hair follicle regeneration. Make sure to also read the comments from Dr. Kellen Chen (one of the FAKI study co-authors) in that post.
See my conversation with one of the Longaker Lab researchers on Twitter in September 2021 regarding both these options. Note that these same scientists (led by Dr. Michael Longaker) published both studies:
- Original Verteporfin study, published in April 2021.
- Original FAK inhibition study, published in September 2021.
Verteporfin and FAK inhibitors both work via the inhibition of the YAP pathway. See part of my conversation with @DrPhil on Discord from last year where he favors the latter over the former:
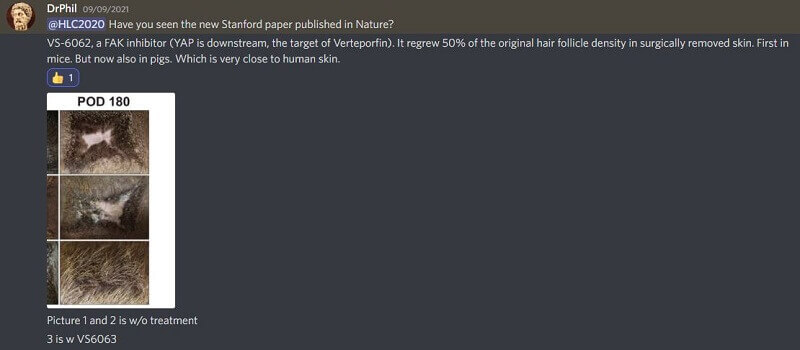
Also per Dr. Phil:
“This VS-6062 FAK inhibitor does the same thing as verteporfin, but it inhibits a target which is upstream of YAP. Verteporfin only inhibits YAP, while VS-6062 inhibits FAK (which controls YAP) and also other targets. FAK inhibitors can also be applied topically, with no injections required. Moreover, VS-6062 has already gone through Phase 1 and Phase 2 clinical trials to treat cancer.”
Verteporfin Human Clinical Trials
The biggest reason for all this recent excitement is due to the fact that a hair transplant surgeon is finally testing Verteporfin in his patients. Verteporfin (trade name Visudyne) is already FDA approved for photodynamic therapy to treat various eye problems.
Therefore, many people were wondering why we need to wait for the Longaker Lab to begin their human trials (after their pig trials finish). After all, any surgeon can test it out off-label right now. Luckily, we finally have our wishes fulfilled. See the next section with videos further below.
While I am not optimistic on any kind of hair regrowth in the donor region after graft extraction, I would not be surprised at some improvement in wound healing. i.e., the potential for hair transplants with minimal donor scarring in the future.
However, it should be noted that most FUE hair transplants seem to result in acceptable levels of scarring. On the other, FUT (strip) hair transplant still often have very obvious signs of scarring (e.g., see Elon Musk’s hair transplant). Also see my poll results on FUE versus FUT hair transplants.
Three new Videos on Verteporfin and Hair Growth
- On June 21, 2022 Hairliciously published a new video on Verteporfin and hair follicle regeneration.
- On June 22, 2022 Hair Transplant Network published a great interview with Dr. Taleb Barghouthi of the Vertex Hair Clinic in Jordan. He is experimenting with using Verteporfin on his hair transplant patients for one month. If the best case scenario is realized, we would see hair regeneration in the extraction sites and a potentially unlimited donor supply. I am very skeptical, but I liked this interview and Dr. Barghouthi sounds very sincere. Even if the strip or FUE hair transplant scars can be improved via the use of Visudyne, it will be a great achievement. So far, Dr. Barghouthi has seen a delay in healing from sites injected with Verteporfin. According to the video description, this could mean that the pathway that signals the body to being scarring is being blocked.
- On May 17, there was an interesting presentation on verteporfin and its anti-scarring effects by Dr. Heather Talbott (who has conducted years of research on wound healing and scarring). Watch the section between 14 mins and 32 mins in the video.
Nasa, what happened to JAK inhibitors being the ‘miracle cure’??
It only works for alopecia aerata not for androgenic alopecia.
At the moment, the main hope is for scar and wound reduction. The hair growth is a potential benefit if the damaged skin regenerates with fully functioning hair follicles and sweat glands. Could work for AGA too per the science, but I would not get my hopes up for humans.
Anthony sent me the below. Interesting.
https://phys.org/news/2022-07-chemical-life-death-hair-follicles.html
Actual study:
https://www.sciencedirect.com/science/article/pii/S0006349522004283
https://newatlas.com/biology/hair-loss-baldness-regulator-protein/
They don’t say if it coulde be useful in the case of Aga… And something tells me that it could triggers cancer risks…. Still far away
https://www.eurekalert.org/news-releases/959702
We are getting there …
They recently discovered protein that tells follicles when to grow/die/regenerate.
Quick question. If you’re completely bald, will these future treatments/cures reverse your hair loss or is this all for people with beginning stages of hair loss ?
They discover this protein every year Alexander. It could be used in the future to cure baldness. I have been hearing this since 2001. Things are no better then and now on their promises……stick big 3 and fue. If it happens it will happen….just don’t wait around for it lol
Finastride, Minoxidil, and ???
A friend jokes about laser caps and Rosemary oil.
https://www.dailymail.co.uk/health/article-11054869/amp/Scientists-chemical-trigger-baldness.html
They keep talking about it … we’ll see
In the meantime my forehead gets bigger and bigger :(
In the meantime is there a way to lower the TGF-beta protein in our scalps somehow?
Let’s say a cure came out today. What would they mean for those who’ve had hair transplants? Obviously the donor area can’t grow back right ? What about the transplanted area? Will that keep us from getting fixed with future treatments?
UT-34 | Androgen Receptor Antagonist/Degrader. Please can someone elaborate about it. Br Kappo.
@josh
I think that hair transplants create scarring in the recipient too and destroy skin structure, so yeah the surface area where hair was transplanted will likely regrow less. That being said imo its debatable if hair loss is reversible past a certain point, also its very possible that the cure will be coming from something like Stemson which creates de novo follicles, and yeah since you can add density right now with transplants too so transplant same area multiple times, that might also worth considering. And of course the most likely scenario that it will be Fin+Minox in 20 years too.
Guys, Dr Baghouti may have cracked something. The pics he posted from his experiment are showing a hair did grow in the extraction area, whether that’s a velus hair or a brand new follicle awaits to be seen, but it is very exciting and we need to have more hair transplant surgeons experiment with verteporfin to see if they can regrow the follicles back after extraction. I am posting a link to the thread for the trial. Very exciting and potentially a game changer. I am cautiously optimistic.
https://www.hairrestorationnetwork.com/topic/64737-verteporfin-hair-regeneration-human-trial-dr-barghouthi-official-thread/page/2/
Wow, Admin.
I remember being a part of this forum back in highschool when my hairloss began. I would wait weeks until something new came about in the hair loss research world. Now it seems like everyday i visit your blog something is breaking grounds ands its getting hard to keep up with.
Im following HMI, SCUBE3 and OLIX more closely.
And still nothing has ever come out. Just endless on-going studies.
FAK Inhibitors sound like another idealistic miracle that will be talked about on here for the next 10 years and never come to anything. Verteporfin seems like it might actually be able to be used in the next 1-3 years.
Endless studies, bald CEOs, happy mices, fancy websites and 0 products… Welcome to 2023.
https://longevity.technology/news/uk-mhra-to-streamline-clinical-trial-approvals-in-significant-overhaul-of-trial-regulation/
This is going to help speed up trials in the UK while maintaining safety/quality standards. Potentially could help any hair related clinical trials there too I think
Great news. They were taking about this many times before (especially after Japan passed similar fast track regulations years ago).
After Covid mRNA vaccines got approved in a year, seems strange to have clinical trials go on for 5 years or more.
This sounds like what Dr. Cooley’s Acell research paper claimed about 15 years ago. Donor scars were said to be nearly invisible with hair growing out of it as a result of applied extracellular matrix. That said, I can claim from firsthand experience that this is false. Also, a PowerPoint from this doctor claimed “autocloning” with plucked hairs had a 70% success rate with microscopic proof. I saw the images. That said, I also know firsthand after trying it that this is false (or we’d all be celebrating my success). The doctor’s website still advertises significant results with these methods. I’m not sure why such a skilled and amazing hairline surgeon needs this type of bologna sandwiches, but the hair loss industry really loves snake oil… https://haircenter.com/cloning-research/ Anyway, I have tremendous caution in my optimism. I hope one day the indisputable real deal comes along :-)
What is wrong on that page? He says the following:
“Currently, there are several companies actively researching ways to multiply hair using cell therapy.”
“While this technology is very exciting and much has been accomplished with animal research, the applications in human clinical studies have proven very frustrating.”
“It appears that this treatment is many years away from becoming a practical reality for patients with hair loss”
“For now, we can’t count on this technology yet as a hair restoration treatment. But it is incredibly exciting research and worth keeping an eye on in the future.”
Usually, anyone who says hair cloning will be here in 2 years is considered to be a liar. Same if they say many decades away. So the above statements seem fairly uncontroversial to me.
Slick—Do you think that autocloning with plucked hairs could work if the plucked hairs had Verteporfin applied in the receipent area. The plucked hairs have stem cells clinging to the root that could start to grow with Verteporfins help.
Hi Admin, just wanted to flag this brief new interview w/ Dr. Mohebi, re: Verteporfin. I think the headline is that he’s starting human clinical trials for it.
This time, with the link: https://www.youtube.com/watch?v=XChdIw3MVHI