I have covered Japanese cosmetics behemoth Shiseido for almost ten years across over a dozen posts. My last post about them became too confusing, as I was regularly appending it with brief new updates. I was awaiting the correct time to restart. This company also makes the popular Shiseido Adenovital Shampoo.
Shiseido’s work on a hair loss cure based on the injection of cultured autologous dermal sheath cup cells (DSCs) is promising and safe. While the company has no problems with fundraising, thay have had some issues with their partnership with Replicel (Canada). I will not repeat that story here, but readers can check out my past posts on those two companies for more details.
Shiseido Phase III Study Results Published
Earlier this month, Shiseido released the results of its “Phase III equivalent” study. The actual work was published in “The Journal Of Dermatology” Volume 50, Issue 10, in December 2023. In that same second link above, it also says “First published: 26 September 2023”, which might mean date of original Japanese submission.
No-one seems to have noticed it on Reddit as far as I can tell. I only learnt about it due to someone e-mailing me and asking me to check the end of the first PubMed link above where it says: “Grants and funding: Shiseido Group.”
It is not surprising that this development went through without notice. For one, the title of the paper does not even mention Shiseido’s name:
“Efficacy of autologous dermal sheath cup cell transplantation in male and female pattern hair loss”.
Even causing more confusion is the fact that they call this a “Phase III equivalent clinical study.” And only 36 volunteers participated in this trial. Not even remotely close to what you expect in a proper large-sized Phase 3 trial. The Shiseido website’s Hair Regenerative Medicine page has no new updates either.
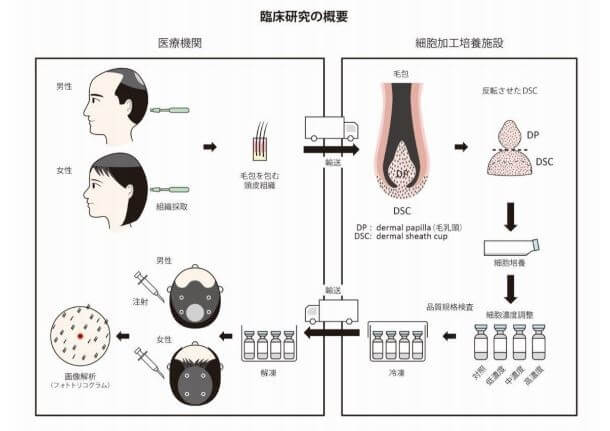
In any event, this is clearly Shiseido’s trial for its DSC injections. After culturing dermal sheath cup cells (the source of dermal papilla cells) from the scalp skin of the hair loss patient, they are implanted (injected) into the same patient’s balding scalp skin to rejuvenate damaged hair follicles and promote healthy hair growth.
Among the paper’s many co-authors are the renowned Shiseido-affiliated Dr. Ryoji Tsuboi and Dr. Manabu Ohyama. All of the co-authors are also associated with at least one of the folllowing four entities in Japan:
- Department of Dermatology, Tokyo Medical University, Tokyo.
- Department of Dermatology, Kyorin University Faculty of Medicine, Tokyo.
- Department of Dermatology, Toho University Ohashi Medical Center, Tokyo,.
- Regenerative Medicine Research & Business. Development Department, Yokohama.
Results
Keeping in mind the small size of the study, the results while positive, are a bit underwhelming. Thirty-six male and female participants with pattern hair loss (PHL) were injected with dermal sheath cup cells. These DSCs were harvested from non-affected occipital hair follicles twice in quarterly intervals.
On global photographic assessment, 30% of the participants showed improvement.
The phototricogram data analysis showed increases in the:
- Cumulative hair diameter of 107.6 ± 152.6 μm/cm2. This was a +1.4% increase versus baseline.
- Hair cross-sectional area of 3069.1 ± 10960.7 μm2 /cm2. This was a +3.4% increase versus baseline.
- Mean hair diameter of 0.9 ± 0.9 μm. This was a +2.2% increase versus baseline.
The female and high terminal hair ratio groups achieved better outcomes. It is not clear if any new hair growth occurred from the above breakouts. But stronger and denser existing hair after 12 months is a positive. And could imply better future protection against dihydrotestosterone (DHT) spurred follicle miniaturization.
Another confusing statement:
Of the total participants, 62.9% noted some degree of improvement.
It seems like this 62.9% figure is based on patient feedback rather than any kind of measurement. And per the math, this would imply that 22 of 35 patients gave a positive response, while 1 dropped out before the 1-year mark.
I have not tried to find any long version of this report, as overall it leaves much to be desired. I hope a far lengthier Phase 3 clinical trial is still going on.
And I also hope that Shiseido will be able to remain fully transparent, in spite of any pending legal issues with Replicel.
It doesn’t sound too promising but super nice work finding this Admin.
Thanks, though someone else sent me the first of the two links and pointed out the Shiseido at the bottom.
Too bad it only worked in 30 percent :-( Hard to believe, after so many years.
Is it? :-(
I would have expected something more like +5% in 70 percent of people.
Ok… So now what? Phase 3 is in, so do they now have a product? What comes next?
You see Moe posted another pic of his scalp (HMI 115)? On his Reddit.
Not a home run but definitely noticable progress.
https://www.reddit.com/r/HMI_115/comments/16qnzgd/comment/kd0iema/?utm_source=share&utm_medium=web2x&context=3
Whoa. The hair trigger fury in that reddit comment section (pun/irony unintended). Some guys react as if they designed the damn thing…
My HMI-115 post was almost 1/3 devoted to “Moeman” and I cut down on that after so many of his past comments on Reddit got deleted. Can’t decide on whether to republish that post.
Is the first first photo in the below thread from three months ago really Mo? I like his second comment:
“I am fat, bald, and near sighted, and I am still liked by friends and family and my wife.”
https://www.reddit.com/r/HMI_115/comments/16vkosl/moe_before_using_hmi_115_his_head_is_completely/
Wow – some decent results (from my perspective).
I agree. If the before and after pictures are legit then this would be the best hair loss remedy on the market.
Mind that he grew back hair on his scalp where he was slick bald for 10 + years – Fin couldn’t do that. It could work even better on patients with lower norwoods.
It’s definitely not a full cure based on Moeman‘s pictures, and I am unsure on how much money I would be willing to spend on HMI-115. A couple of thousand bucks, yes sure. 10s of thousand, nope.
I am too lazy to check, but I think Moeman got the same dose as the macaques (120 mg per injection). There was a thread on Reddit where multiple users argued that those monkeys only weigh around 10 kg and a dose for humans would need to be much higher in concentration in order to be as effective. I‘m really no expert in antibody-therapy and therefore don’t know if antibodies work that way or if that could be tweaked by more injections. Maybe somebody in the forum can jump in and lecture us idiots.
In phase 2 there‘s a trial cohort which is treated with 240 mg/injection – so there’s that.
Agree to disagree – if we’re looking at the same pics (I see two photos and can’t tell if they’re before or after but in neither do I see significant hair – he’s bald in both).
I imagine this isn’t going to be cheap (and injecting stuff into you is pretty serious – mess around and wind up with a third butt cheek) so yeah it’s a no from me. Not a cure and definitely falls short of all the claims and all the hype. If I still consider myself bald afterwards, I’m not interested. Unless it’s dirt cheap. Maybe then I’d think on it. But we all know it won’t be cheap.
Thanks, Admin – I’m of the mind that anything forwards, even incremental, is a positive step. Any time you post a new post, my hopes receive a slight renewal. Definitely not a bad thing. Appreciate all your hard work, and Happy New Year.
Thanks, and same to you Kyle.
Hello Kyle..hope is good. But it can be a dangerous trap. I am convinced that many guys cling on hope and not using existing treatments due to fear of side effects. They wait and wait and wait..in the meantime they losing more hair. Once they realize that hope is not a solution they go to the butchers..many in Turkey. Then they start with existing treatments. Because if you do not after a HT you are a complete fool IMO.
The trolls have done a good job!
Woofy is a good example…Woofy am I right?
Hey, MRKA… I hear you, good buddy — but I think it depends on one’s perspective, and how connected this issue is to one’s self-image. While I hate that I don’t have the density I once had (especially as I always shaved my head in my 20s and 30s, so ironically never took advantage of the thick, wavy hair I had), I like everything else about myself so am ultimately happy as I am — benefits of being an egomaniac, haha. It doesn’t stop me from seeing myself the way I want to, and I find it easy to keep it all in perspective. So for me, it’s not nearly important enough to get a transplant or take the risks of certain side effects (which again is easy for me, as I have a natural low risk tolerance outlook). I just put my energy/focus into other things, and trust that if I ever change my view it’ll be when I’m too old to care.
Agreed!
Disappointing results but honestly I wasn’t expecting much. Too many delays makes you wonder (and doubt). It’s unfortunate but not surprising. Another one bites the dust. We need to stop getting our hopes up too much. When we do we get crushed with results like this (e.g., some of the Reddit folks big mad).
I agree that any gains are good but I don’t want to have to use 25 products (pills, shots, shampoos, topicals etc). A few okay, but I’m also not paying tons of money for minimal gains. If I’m still considering myself bald afterwards, it ain’t worth it to me to go in for some procedure like this.
But of course it’s a personal decision.
FYI — I have an RSS feed link on the site, but some people have complained about it in the past. Just realized that there is another option for people who use Chrome:
https://9to5google.com/2023/02/10/chrome-follow-feature/
I think Moeman’s pics, if real, are pretty encouraging. I’ve never seen fin or minox give that level of growth. Not perfect, but he got a lot of hair. And, would he have more growth if he got larger doses, or if he continued getting shots every two weeks?
I agree I think moemans results are fantastic! Getting a shot with no bad side effects every few weeks and not having to deal with crazy fin sides or minox bloating?! Heck yea sign me up!…and Shiseido needs to actually follow thru with this. Even if it’s a maintenance treatment, many, including myself, would be interested. . Like cmon, either poop or get off the pot.
Hey Admin,
Hope everything is well with you and your family during the busy holiday season. With these lackluster results from Shiseido, are your hopes for an imminent new treatment/cure now extinguished? I believed that Shiseido gave us our best hopes at any cure/treatment on a more reasonable timeline. Now that this is not the case, is it reasonable to expect any treatment to come to our rescue any time soon? Hair loss is a horrible disease/affliction and I wouldn’t wish it on anyone.
There’s still Yokohama, Epibiotech and Amplifica all with potentially better timelines (maybe Pelage).
Thank you Yoyo :)
Do you think there’s a chance that these could come to the market soon? Heck, I’d be happy just to know that there’s something that WORKS. Most of these therapies in development don’t even offer great results.
It appears that Shiseido and Epibiotech’s EPI001 follows the same approach that is injecting the cultured DSCs. Since Shiseido didn’t get the desired result should we remain hopeful for EPI001? When CEO Sung himself previously responded in comment of some previous post that DPC may not be effective as transplantation for severe hair loss patient.
No worries. It’s difficult to say with certainty, but with the more attention the hair loss area is getting, it is definitely a step in the right direction. Companies like Yokohama and Epibitech are companies based in Japan and South K. respectively so their regulations won’t nearly be as tedious as the ones in the US. If all goes to plan maybe 2026-2027? With Amplifica if the science checks out (which it seems to be from what I’ve read) it’s effective. I wouldn’t be surprised if it could potentially come to market in 2026. I’m not sure where Pelage stands.
Pelage must be the greatest mystery in the industry…what are they doing with their 16 million $ of funding?
I hope they finished their first trial already, otherwise it would be disappointing if they have yet to initiate human tests.
I mean, it‘s one molecule (or 2 in this case) which they determined 6 years ago.
The other blog said they had an interesting scoop on Pelage last month, but nothing ever materialised.
I know admin here thinks niostem is a non-starter, but they appear to be targeting the same pathway as Pelage, just with electrical rather than chemical stimulation…
I never said non-starter. I even wrote a post about the company! But regrowing in a totally bald area is a non-starter.
Fair enough, I seemed to remember a post where you said you were skeptical about laser, mechanical and electrical approaches to AGA working.
I am optimistic about most such things being able to thicken existing hair. We hope they can regenerate some shrunken vellus (formerly terminal) hair too. Wounding sounded especially promising in creating new hair, but alas Follica.
Yea I found this a few days ago Ben, it’s the first news I’ve heard from Pelage (at all). https://www.linkedin.com/posts/octane-oc_hairrestoration-aestheticsinnovation-atf2024-activity-7141510822077390848-2KEX?utm_source=share&utm_medium=member_desktop
Ok thanks YoYo…I guess we have to wait for this congress then to get more news from them.
Very poor results unfortunately. Thanks for the update, Admin.
Histogen will be the final cure in 2024.