How does Minoxidil work in growing hair on a receding hairline? I regularly update this post with newer studies discussing this topical drug’s mechanism of action in stimulating hair growth.
Minoxidil (brand name Rogaine) is known to be one of only two medications officially approved for hair loss treatment. With the other being Finasteride (Propecia). It is available in 2% and 5% dosages and in topical and foam versions. Besides Rogaine, another well known Minoxidil brand is Costco’s Kirkland.
Minoxidil was first approved by the US FDA to treat male pattern baldness (MPB) in 1988. In 1991, the product was also made available for women with female pattern hair loss (FPHL) and thinning hair.
Make sure to read my post on whether Minoxidil can grow a beard. A large number of people are using this product to gain facial hair, not something I would recommend. Also see some before and after results of Minoxidil and Finasteride to regrow hair.
How does Minoxidil Work?
Scientists do not know the exact mechanism via which Minoxidil® (a mitogenic drug) has a positive effect on hair growth. However, there are a number of proven mechanisms of action that suggests how Minoxidil works to stimulate hair growth.
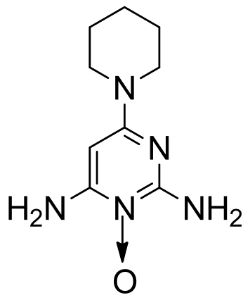
The original use of Minoxidil was as an oral medication for high blood pressure. See my post on oral Minoxidil. The side effect of hypertrichosis (excessive body hair) led to its becoming a popular treatment option for hair loss.
To date, the main theories about how Minoxidil works to grow hair are:
- Due to its proliferative and vasodilatory actions.
- Due to its potassium channel opening effect.
- Via inhibiting the decrease in calcium levels in cells. This prevents the inhibition of epidermal growth factor stimulated hair root growth.
- By increasing blood flow to the scalp.
- By increasing key growth factors that are conducive to scalp hair growth.
- Prolonging the anagen phase and decreasing the telogen phase of the hair cycle.
There are also other theories about how Minoxidil (aka Rogaine) helps scalp hair growth. Further below, I outline all of the main ones. Note that some people use Rogaine once per day rather than twice per day.
Besides prolonging the growth phase of the hair cycle, Minoxidil has also been shows to increase the diameter of existing hair follicles. According to a study from 1988, seven subjects who received a 5 percent dose of Minoxidil had a mean hair shaft diameter of 0.029 mm before treatment. This hair width then increased to 0.043 mm at 12 weeks.
It should be noted that Minoxidil can grow hair on both the crown (vertex) and frontal regions of the scalp. Many people think that it only works in the crown area, but that is incorrect.
Brands
Minoxidil’s original brand-name is Rogaine. However, after Upjohn’s patent ran out, a number of generic brands have come into fruition. The most popular of these are Walmart’s Equate brand and Costco’s Kirkland brand. Note that both come in topical solution versions as as well as foam versions. Also check Walmart’s site as well as Costco’s site for oftentimes better pricing.

Also popular is Lipogaine, which contains a Minoxidil 5% solution along with: Biotinyl-tripeptide; Niacin; and Apple polyphenols for stimulating hair growth.
History of Minoxidil
- In 1979, Minoxidil was first approved by the US FDA. However, its usage was as an oral blood pressure medication (brand name Loniten) rather than as a hair loss treatment.
- In 1988, the FDA first approved Minoxidil as a hair loss treatment for male pattern baldness (aka androgenetic alopecia). This was via a topical 2% solution formulation under the brand name Rogaine, owned by Upjohn Company. However, Rogaine was only available via prescription, and it was only meant for use by men.
- In 1991, the FDA approved Minoxidil as a hair loss treatment for women, but by prescription only.
- In 1992, Rogaine launched a 2% Minoxidil topical solution for women, only available via a prescription.
- In 1996, the FDA approved the over-the-counter sale of 2% topical Minoxidil and also allowed the production of generic formulations of the product. Upjohn subsequently drastically cut the price of Rogaine in order to compete in the now much more competitive market.
- In 1997, Upjohn released a 5 percent topical solution formulation of Rogaine. It was only available by prescription and for men only.
- In 1998, the FDA approved a 5 percent formulation of Minoxidil for over-the-counter sale to men.
- In 2006, Rogaine launched a 5% Minoxidil foam product for the first time. It was immediately available for over-the-counter purchase, but only recommended for men.
- In 2011, men’s Rogaine foam was made available in unscented format.
- In 2014, Women’s Rogaine 5% Minoxidil foam was approved by the FDA, but with a once a day treatment recommendation. Health Canada also approved the 5% for FPHL. Men are always supposed to use the product twice a day.
Mechanism of Action (References and Studies):
- In 1997, researchers found that Minoxidil increased prostaglandin synthesis (more specifically, prostaglandin synthase-1, abbreviated as PGHS-1) in cultured dermal papilla cells. In more recent years, the issue of prostaglandins and hair loss has garnered a great deal of attention and you can search for “PGE2” on this blog to learn more.
- A French study from 1998 is among many that has found that Minoxidil upregulates growth factors, in particular vascular endothelial growth factor (VEGF).
- A 2001 study found that the positive effect of Minoxidil on hair is mediated by adenosine.
- A 2004 report suggests that vasodilation, potassium channel opening and cell proliferation plays a major role in the mode of action of Minoxidil.
- An excellent article from 2008 on hair loss medical treatments by Dr. Nicole Rogers and Dr. Marc Avram that discusses Minoxidil in detail. They mention that one of the main effects of Rogaine is angiogenesis and increased blood flow in the area of application. They also discuss the enhanced cell proliferation and DNA synthesis effects on Minoxidol that might be benefiting hair growth.
- In 2011, South Korean researchers found that Minoxidil activated the β-catenin pathway in human dermal papilla cells and therefore extended the anagen (growth) phase of the hair cycle.
- In April 2014, Taiwanese researchers came up with yet another reason as to why Minoxidil works, concluding that it may suppress androgen receptor-related functions. i.e., the drug has anti-androgenic properties. Their conclusion is especially interesting:
The current findings provide evidence that minoxidil could be used to treat both cancer and age-related disease, and open a new avenue for applications of minoxidil in treating androgen-AR pathway-related diseases.
- In January 2017, Turkish scientists concluded that Minoxidil acts as an antiandrogen.
- In February 2017, an unrelated study from Japan had this to say about Minoxidil:
Minoxidil enhances hair keratinocyte proliferation and activates hDP cells to induce growth factors. IGF-1 is among these growth factors, and has been shown to exhibit a potent hair elongation effect.
- In June 2017, US researchers published a new study that provided significant insights into how Minoxidil foam worked via upregulating and downregulating various genes. Interestingly, vertex and frontal scalp of patients showed a generally similar response to Minoxidil. Many online reports suggest that it might work better in the crown than in the front, but perhaps that is not true based on these findings.
- In February 2018, South Korean scientists discovered that Minoxidil promotes hair growth through the stimulation of growth factor release from adipose-derived stem cells. This growth factor secretion may enhance hair growth by promoting dermal papilla cell proliferation.
Minoxidil Side Effects
In general, topical Minoxidil is well tolerated in most people at the typical 5% dosage. Most people even tolerate higher concentration levels of the drug. However, some people will get side effects.
The most common entail adverse skin reactions such as burning, itching, redness and stinging in the areas of application. Another common complaint is an increase in body hair growth after taking Minoxidil, especially in the forehead, eyebrow and beard regions.
In rare instances, people complain about dizziness or breathing difficulties after taking Minoxidil. Allergic reactions, including rashes, are also possible in some cases. Please see a doctor immediately if you get such serious side effects. Also make sure to stop using this medication right away.
Minoxidil Wrinkles and Dark Circles
Various online reports from users suggest that topical Minoxidil can cause wrinkles and facial skin aging due to loss of collagen. Some people also complain about increased dark circles. However, there are no major studies that conclude that Minoxidil can cause wrinkles or dark circles.
The evidence of facial structure and collagen synthesis changing from Minoxidil use is just not there at present. Of course if you are using the liquid version of the product and it regularly pours down your face, unusual side effects are always possible. I prefer the easier-to-use foam version of the product myself.
Rogaine Shedding
Some people will shed a lot of hair after changing their Rogaine (Minoxidil) dosage. For those who quit Rogaine entirely, a major shed of scalp hair is almost always guaranteed. Sometimes this can take weeks or even months after drug use cessation. In many instances, sheds are temporary and just a regular part of the hair growth cycle. i.e., anagen, catagen, kenogen and tologen.
Minoxidil Sulfate and SULT1A1
The Minoxidil sulfotransferase (SULT1A1) enzyme booster enhances hair regrowth from topical Minoxidil. Also, in May 2020, Japanese scientists concluded that arylsulfatase inhibitors may sustain the effect of Minoxidil sulfate in androgenetic alopecia therapy.
Toxicity in Cats and Dogs
If you own pets, note that Minoxidil is very poisonous to some animals, especially cats. See this sad post on Reddit regarding Minoxidil killing a person’s cat.
If your cat (or dog) is exposed to Minoxidil via a spill or accident, some side effects to look out for include:
- Fatigue and lethargy.
- Changes in heart rate due to cardiac damage.
- Dehydration.
- A drop blood pressure (hypotension).
- Coughing.
- Changes in appetite.
Prompt action and treatment by a veterinarian will prevent your cat from dying. If the medication was applied topically, make sure to wash the cat’s paws and fur promptly and thoroughly.
Hi
I had hair loss a couple years ago due to some underlying issue, which has now been resolved.
I resorted to Minoxidil immediately (which in hindsight I wish I hadnt), and my hair began to shed like crazy.
I stopped using for a month, and almost a year later my hair has not recovered from the shedding period.
The underlying issue is no longer present, I have no real hair loss, and no miniturisation of the hair shafts.
Can you shed some light as to why this might be?
Minoxidil is undoubtedly an effective treatment until it stops use ; as if you have not used a cure ; Minoxidil is also effective in some men and women and is negative with them too; I see that it is not a cure but it repairs the damage.
We are waiting for a real treat.
Regarding the risk to cats, what about indirect exposures?
For example, If you apply rogaine, wash your hands but minutes later you put your hands on your head and then put these hands on the cat? Or If the cat lick your hairs or even the pillow you use to sleep.
Can the cat be intoxicated?
As far as I’m concerned, Minoxidil is the only thing in existence that has a good shot at regrowing significant portions of your hair. It has been an absolute life-saver for me, and I’m only I’d say about an average responder. Best part is that it’s relatively cheap, and has no real side effects (for me). For people that respond incredibly well, it effectively reverses slick baldness.
I’m a good responder. I was a early NW 2.5 (Hair line & Vertex). Used it in May 2019 and now… a NW 1.5 (straight hairline and 90% filled out vertex). Still regrowing hair interestingly.
@admin in your option, for folks who just started maintaining early hair loss (Nw 1.5-2.5), do you suspect (and if approved) a combo of Follica (follicle Neo genesis) and Breezula (halts hair loss) to essentially be a “cure” for this subgroup ? And obviously a transplant for those who want a Ronald Regan NW 1 hairline.
Hi Handsome Squidward,
That sounds correct, although I will only truly believe Follica’s efficacy after many users give their ratings and opinions on it.
Minoxidil has been good to me. I just use it and nizoral and have done for 5 years. Still losing ground but thankfully at much slower rate. Perhaps had a slight impact on dark puffy circles under eyes but that could just be genetic, some friends have it way worse and don’t use it. I’m knocking on the door of 30 now. Started at 24 and glad I did. The foam works great and absorbs fast. Takes 2 mins in AM and PM. I would recommend to anyone.
Random topic and hopefully it doesn’t hurt the topic of minoxidil too much, but I am very sad and I am trying to figure out if my hairloss could be fungal related?
I have MPB style hairloss, a lot of the sides/back top has thinned out aggressively, the front middle has decent thickness but its clearly thinning out too. Was wondering if it could be fungal related because I do have acne and I have acne spots on my upper chest/upperarms/back which is a sign that my acne problems could be related to malassezia (fungus).
When I am running outside, I have moderate itching when I get super sweaty not only on my scalp but my face and neck area as well. I did take antibiotics when I was younger for my acne(minocycline) which POTENTIALLY might have made the fungus more resistant which could potentially have triggered these issues (mpb/redness of face/itching)
Any opinions about this?
Cut out all forms of dairy and your acne will disappear. Worked for me.
I did read many years ago a hair specialist sayn to a patient whether his hair loss was due to fungal problems or was it sugar related but what he meant was not explained.
When I was a teenager I suffered acne. I use to bath and washed my hair with soap. Could not afford shampoo. I remember one time during a bath that my hair became tangled which never happened before. It was falling out in the bath and I could see roots connected to the hair follicles. I ran my fingers through my scalp and there was this horrible thick gunk on my scalp. I believe it now to have been soap residue gunk. I went straight to my barber and had all my hair shaved off. I went to chemist and got some sort of dandruff medication to wash my scalp. I went to school wearing a beanie. Later one morning I woke and there was all this hair on my pillow, I could not stop it. Later I suffered Male Pattern Baldness.
There is a theory that soap scum blokes hair follicles and looking back, I believe this to be true. I also was in the back of a convertible and my hair was getting blown about. We drove for many hours. The next morning, I could not comb my hair because of the pain. They say that you can damage hair follicles so I also wonder if I did irrepairable damage and I wondered how motor cycle riders go on in the early days when helmets were not mandatory.
I did read where this bloke did autopsies on car accident victims. Some were scalped. This person noted that men who hair healthy hair had clean scalps undreneath whereas men who were bald had gunk under their scalp indicating blocked hair follicles. Thts why I believe soap scum may have blocked my hair follicles but am only guessing.
To back this hypothesis up, there is now a product running along this line that baldness is due to blocked hair follicles and by using their product it will cleanse hair follicles and allow hair to grow again. Its called Fotero.
Well thats my story.
But baldness is not good.
Interesting study on Oral minoxidil monotherapy
https://www.dermatologyadvisor.com/home/topics/general-dermatology/oral-minoxidil-possibly-effective-for-male-androgenetic-alopecia/
Off topic but I thought noteworthy that Frequency Theraputics pulled in another $62 million in venture capital on top of the $80 plus million they got last week from Astellas. This seemingly adds weight to their progenitor cell regeneration model with the hope that scalp hair isn’t far behind in their research timeline.
Funds also to be used to “….advance discovery programs in other therapeutic areas….” The Frequency website mentions that AA is at the top of that list. Here is the link the article: https://www.businesswire.com/news/home/20190723005152/en/Frequency-Therapeutics-Completes-62-Million-Series-C-Financing
Seems promising, but their main project is on hearing loss… My bald and balding fellows I’m only 20 years old with severe diffuse AGA and starting to lose hope already. So many breakthroughs, still nothing though.
This won’t be the next possibility out there, but at 20, this is a solid development. Their platform is attracting a lot of capital: “We were impressed by …….the elegance and the underlying science of their platform…” https://www.drugdeliverybusiness.com/frequency-therapeutics-inks-625m-deal-with-astellas-adds-62m-series-c/ While you are correct that their current trials are for regenerating ear hair, the latest funding will be used to develop other therapeutic uses and they are already researching a treatment for androgenic alopecia. See PCA Regeneration Opportunities https://www.frequencytx.com/our-approach/pca-regeneration-opportunities.php
Even at 20, as you probably know by now, progress moves painfully slow in this arena. But relatively speaking, Frequency seems to be moving quickly. They just came on the radar in February of 2018, and by April of this year, they had already announced positive Phase 1/2 trial results although I’m not sure what a 1/2 trial is. Phase 2 to commence this year. Theoretically, this could pave the way for similarly quick development of an androgenic alopecia treatment, research on which has already been ongoing for the past year. So as possibilities go, I personally rate this one high.
GreedBlocksTheCure … Your name shows that you have a real understanding of how establishment money makers will aggressively squelch progress on any threats. That said, you’re lucky to be so young, with just a few years left until a real cure. Guys are in physical prime until age 35, so you have plenty of time.
Right now, I recommend shaving your hair short. There are incredible SMP artists out there to supplement with a subtle texture (to avoid horse shoe pattern and maintain your youth). You can get FUE transplant to supplement, and it’ll never limit your future options to clip hair short or use the future “cure”. Never ever get linear strip scars (FUT)! That’ll limit your ability to clip your hair short and sucks terribly for very bald people (who start losing hair at 20).
That’s all the advice I got. Just clip it short, get some sun, work out, and enjoy life. SMP can help you keep a young hairline illusion (highly recommended finding a good artist.. I know a guy). Good luck!
Thanks for the advice mate, really appreciated. But, I think I’ll pass by on SMP cause it looks unnatural on most people and you turn into a play-mobile ( my opinion ). Concerning FUE, it seems like a good idea once I turn 26. Let’s hope till then that something else has come on the surface.
SMP is extremely effective if you have a good artist who does a subtle job with a realistic irregular hairline. I’ve had 4 hair transplant surgeries and hair systems, but modern fine-point SMP looks the most natural by far for keeping a young and natural look (helps that my hair transplants give it 3D stubble). The key is to be subtle so that hardly anyone notices, yet they just think you shaved your hair as a choice. Anyway good luck!
My greatest concern with Minoxidil are the old studies that showed an increase in Left Ventricular Mass with use of 2% topical Minoxidil. To my knowledge no further studies have been done on the subject. Here is one of the old studies:
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1386573/
Do someone have any more knowledge about studies on Minoxidil and heart health? Its hard for me to understand why the 1988 study was not followed up by studies with longer duration.
What happened to the Italian lotion (don’t remember the name) that everyone was waiting for and was hyped a few months ago? Any update on that? Anyone seen result?
The name of the lotion I mean is Trinov.. It was really hyped and not seen a word about it since anyone knows what about it?
I think you’ve answered your own question. 2019 has seen Brotzu and Aclaris throw down the gauntlet. Soon to be Shiseido. I was originally very optimistic but their delayed release of the results has me sceptical.
@Scott what got me really sceptical is the dispute between shiseido and RepliCel. They either have a real breakthrough and shiseido ( as the big player ) wanted to get RepliCel off the game through the legal path, or got nothing so they buy time by delaying the results release.
Puretech says Follica is going to finish the pivotal phase with a FDA filing during 2020. Moreover says that Follica is one of they key value drivers.
What does ‘value driver’ mean
something the company expects a lot of money from
Ah, I see. So Follica is a nested business in Puretech?
Yes, you can check it on puretech’s website
This link doesn’t work:(
http://puretechhealth.com/images/PureTech-Corporate-Presentation_FINAL-7.25.19.pdf
Working Link:)
If they delay this pivotal trial one more time for this optimization study I’m going to go crazy lol!
Pier, The link doesn’t work. I went to the investor presentation tab (reports and presentations) and there is 7/23/19 report but I do not see the comments you mention and page 21 deals with LYT-210. Can you check your link?
My apologies if this is the wrong place to post this information. But I believe its significant and wanted to share . I have HMO insurance and seeing my dermatologist for AGA (Loyola clinic in Chicago). The clinic is now performing PRP shots, which is new procedure for them. My insurance only covered the scalp biopsy , but the dermatologist performs 6 session PRP shots for 1500 (~250 per session) which they perform at the Loyola clinic. Hopefully, the insurance companies eventually agree to pick up the tab for PRP sessions in the future.
I have had it done and know others who have had it done. For sports injuries it’s amazing. For hair loss, it’s useless. Save your money and wait for something whereby you can tell the difference without a microscope.
My 2 cents:
Pontus, you have some very good concerns with minoxidil. I have wondered the same. Could it be that if they uncovered more minox bad side effects that there would be a major major law suit beyond what anyone has ever seen?
Eddie, insurance companies will never approve to cover prp. Sorry man, not gonna happen. It’s an aesthetic thing. That would be like insurance companies paying for rhinoplasty. Not happening.
I’m less impressed with follica and puretech every day. Not only are they taking forever, but their technology/invention is based on current treatments. If you dont believe me, just look on their web site. All they are doing is giving you some new hair that you can maintain with the current treatments.
I enjoy coming to this site however one thing I would like to see change is the small number of people that respond to others questions or statements. How many of you passed up on responding to Eddie’s and pontus? You knew the answers or info yet you just pass them cause it doesnt suit your needs. It gets annoying when you throw a question out there and you get skipped over by the community, I’m sure.
I like to respond if I think I can save someone a lot of money by telling them my experience doing the same thing.
And I don’t do it to act superior, because after all I was the original idiot that did the PRP / laser cap / etc. I wasted thousands and I still look like the villain from the second blade movie.
Note, this article was published only a few hours and rose straight to the most-read—There is HUNGER!
https://www.theatlantic.com/health/archive/2019/07/hair-for-all/594826/
Nice!
Who cares if follica uses current methods. If it can grow 30cm2 hairs than bring it. So you would rather wait another decade for something revolutionary to come out like tsuji and still be banding all those years? I don’t get some people’s logic. If they can find a more effective way of wounding and minox cocktail that grows new hair and thickens existing then they got a good product. They are taking forever because they have to go through all the bs redtape. I’m rooting For follica. That other company focusing on hair loss sounds great but they don’t focus on hairloss. It’s just polaryte with their skin growing. They said hair loss is next…….two years later they went quiet. It’s all about funding and boosting awareness…same circle
Top 2 new treatments that has a chance for release
Follica
Samumed
Everything is a decade away for commercial use or will fail…
And breezula u forgot! Thats actually the one im most excited about for the near term, i.e nxt 2 yrs
What happen Tsuji? Sorry haven’t really been a frequent like I use to.
He is still on time as far as his schedule goes, but he shouldn’t be expecting new results from him until mid next year at the absolute earliest.
I agree with mjones christiano hairclone polarity terskikh all these companies are 5 years away.
I believe tsuji will be on time however I think it couldl be expensive. As for shiseido we should be finding out within weeks. Follica is best deal yet.
Mjones they said 100 terminals per cm2 or 30?
Egghead- follica said 30cm2 terminal hairs and 100cm2 neogenic. Not sure what neogenic is…hopefully new created hairs that go terminal with continued used of their cocktail. Either way 30 new terminal plus thickening of miniaturized hairs is great. It will do wonders for my diffuse loss and those with nw1 to 4. It will help those higher norwoods grow hair and be acceptable for ht.
where you find these data? Any press release about that?
You can find what they mean for neogenic in their website, it’s so unique.
MJones have you ever given thought that many people cant use fin or minox? That they react badly to a prostrate shrinking drug and heart medication?! Of course we want something other then something that amplifies current treatments. Commonsense. .