 Read the bottom half of this post for a history of Hope Medicine and Bayer’s HMI-115. This treatment targets the prolactin (PRL) receptor to cure hair loss. Hope Medicine raised $56 million in funding in 2021. See their pipeline page for more information.
Read the bottom half of this post for a history of Hope Medicine and Bayer’s HMI-115. This treatment targets the prolactin (PRL) receptor to cure hair loss. Hope Medicine raised $56 million in funding in 2021. See their pipeline page for more information.
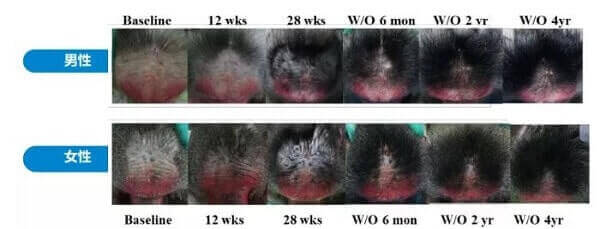
The initial reason for all this excitement was due to the spectacular long-lasting hair growth results seen in stump-tailed macaque monkeys. These monkeys are still being monitored and have not lost their hair even over four years after stopping prolactin receptor antibody treatment.
Update: January 14, 2024
Positive Outcome for HMI-115 in Phase 1b Study in Australia
Really excellent news to start of 2024. The Phase 1b study for HMI-115 in Australia showed positive results in terms of efficacy and safety. The study included 12 males and 4 females. It seems like “moeman32” from Reddit was not lying and I am glad that I covered him for months despite his erratic behavior. See the Australian participant section further below for more. Key quote from this latest press release:
Specifically, in the 12 male patients at the end of the trial, the mean non-vellus target area hair count (TAHC) increased by 14 hairs/cm2, compared to that of the baseline. This increase is statistically significant.
This study is the first ever to show that prolactin receptor blockade can promote hair growth in patients with androgenic alopecia and may provide a novel therapeutic approach.
We also got an update from CEO Nathan Chen that the Chinese Phase 2 trial recruitment of 180 patients is ongoing. A bit slow considering that they started this process in August 2023. It seems like they were awaiting the final results of the Australian trial before proceeding further in China. They aim to achieve full PoC (proof-of-concept) by the end of 2024. No updates on the delayed Phase 2 US trials.
Update: October 27, 2023
Hope Medicine and Chime Biologics Enter Manufacturing Agreement
Hope Medicine (China) and Chime Biologics (China) have partnered to “speed up” the manufacture of the first-in-class monoclonal antibody drug HMI-115. Chime Biologics is a leading contract development and manufacturing organization (CDMO). The two companies are committed to commercialization of HMI-115 for the treatment of both androgenetic alopecia and endometriosis.
Update: August 1, 2023
Phase 2 Trial Recruitment Begins in China
The HMI-115 Phase II clinical trial recruitment for androgenetic alopecia in China has now started.
Encouraging Updates from Australian Phase 1 Trial Participant
A November 2, 2022 detailed Reddit update from an Australian trial participant (HMI-115 dose 1 and dose 2) is required reading. This 37-year old male described in detail his first two doses of the prolactin blocker via injection. He (along with his 46-year old female partner) will get treatment every two weeks for six months. So a total of 12 doses, all at Sinclair Dermatology in Melbourne. Note that while these are Phase 1 trials in Australia, the same HMI-115 product is scheduled to undergo Phase 2 clinical trials in the US anytime soon.
This Reddit user “moeman32” seems legitimate and has a posting history of over ten years. He has also written about Australia many times in the past. He will do follow-up posts every two weeks. Note that all 20 patients in this Phase 1 safety trial are getting the actual drug injected. i.e., no placebo participants needed to test for efficacy this time around.
Update February 8, 2023: Unfortunately, he had to remove his posts due to some stalker issue. But the below threads have as yet not been deleted.
- The beginning (dose 1 and dose 2).
- First follow-up (dose 3).
- A really encouraging second follow-up (dose 4).
- Third follow-up (dose 5). Another positive update.
- Fourth follow-up (dose 7). Seems like there was a miscount in dose numbers and we added one. In any event, I have no reason so far to doubt this person’s honesty.
- Fifth follow-up (dose 8). Perhaps the most encouraging one to date! Just in time for Christmas.
- Sixth follow-up (dose 9). They showed before and after photos for the first time.
Update: April 12, 2022 — Australian Phase 1 trials for Hope Medicine’s HMI-115 are starting today. They will involve 20 participants and are expected to be completed in July 2023. The trials will be led by the renowned hair loss expert Dr. Rodney Sinclair. And there is lots of excitement on Reddit.
Meanwhile, US Phase 2 trials should start at any moment now (read further below). This difference in trial progression by country is strange. Perhaps is has to do with some stricter Australian government requirements.
Update: January 26, 2022
Hope Medicine HMI-115 US Phase 2 Trials
Hope Medicine just received FDA approval to commence Phase 2 clinical trials in the US for its HMI-115 product to treat androgenetic alopecia. HMI-115 is a human monoclonal antibody drug that targets the PRL receptor for the treatment of male and female pattern hair loss. Last year, the US FDA also approved Phase 2 trials of the same drug to treat endometriosis.
The Phase II clinical trial of HMI-115 for the treatment of androgenetic alopecia:
“Will be an international multi-center, randomized, double-blind, placebo-controlled study. It will be carried out in the United States, Australia and other countries.”
Update: November 28, 2021
New CEO Henri Doods Interview
A reader sent me the below screenshot of Hope Medicine CEO Henri Doods’ Chinese magazine interview that I mentioned earlier. If anyone can translate into English, please post in the comments. Click on the image to expand.

Update: September 20, 2021

Reader “Karl” just notified us that there is a brand new cover page interview of Hope Medicine’s CEO Dr. Henri Doods in a Chinese magazine “tradetree.cn”. If anyone can gain access to it and translate into English, please try. Not worth purchasing as yet in my opinion.
Per Karl, Phase 2 clinical trials for hair loss product HMI-115 are planned for Q4 2021 (but no proof anywhere else). This matches what we expected per the prior updates that I discussed in May (see further below). Note that HMI-115 is the same as Bayer’s prolactin receptor antibody. Hope Medicine has the rights to the development of this product.
May 9, 2021
Hope Medicine (China) Raises $56 Million
Until now, Kintor Pharmaceutical (China) was the main reason for recent excitement. However, this weekend, the participants on this site’s hair loss chat are discussing Hope Medicine. Out of the blue, this startup company received $56 million in a Series B round of financing. Investment firms Qiming Venture Partners and Grand Flight Investment led the way.
Other investors include: HighLight Capital; Sinovation Ventures (a venture capital firm led by former Google China head Kai-Fu Lee); and Trustbridge. More here.
HMI-115 PRL Receptor Targeting: Bayer License
“Previously, HopeMed signed an exclusive license agreement with Bayer AG on the development and commercialization of a human antibody (HMI-115) targeting the prolactin (PRL) receptor for the treatment of male and female pattern hair loss. At present, HMI-115 has completed phase I clinical trials in the European Union, with positive safety results. The global multi-center phase II clinical trials for androgenic alopecia and will soon be launched.“

Note that I first covered Hope Medicine in my 2019 post on its partnership with Bayer in regards to the prolactin receptor. The company was founded by Dr. Rui-Ping Xiao, the dean of the College of Future Technology of Peking University. Apparently, she is also an associate editor of the New England Journal of Medicine. Update: See this page for much more on Dr. Xiao Ruiping.
The most unusual part of this story is that Hope Medicine’s official website (hopemedinc.com) has not worked for weeks.
Dr. Xiao Ruiping’s personal story:
https://www.youtube.com/watch?v=hKuWx8OpY_Y
The number of different approaches is amazing: cell-culturing, dht/hormones, antibodies, cell-signaling pathways, wounding, gene/Crispr, prostaglandines, lactate,…
I hope something comes to fruition.
Btw Stemore of Korea also works on antibodies. They renamed their company: “Epibiotech” – I always wonder why that is necessary as they don’t market a product (yet).
There are a lot of moves happening in South Korea that we don’t know about yet, I speculate
Yes and all works on mouses and monkeys.
I notice that topical finasteride named “Caretopic” is out in Italy, not galenic version, it’s an industrial versione.
@Pier Caretopic is the Brand of Almirall / Polichem … do you have a link where to buy it?
Great article and progress in hair loss treatment angles. Nice too see so many different approaches to fight mpb. Sad thing is we have a 99.9% chance these all fail and we are just stuck with big 3 again. Makes you think what is going on behind the scenes.
I’m really excited to see some of the long term results of this treatment. I always guessed DHT was a reason for hair miniaturization but was not the sole culprit. Fingers crossed on this one. It will certainly be expensive. Is it an injection that will be required periodically life long? Or for a certain period of time?
They monkeys didnt show any sign of losing their hair 4 years after treatment ceased, it might be once a decade at worst if it actually works.
Tick tock, tick tock, tick tock…(sigh)
Tired of painting my scalp with dermach every morning.. can we please cure this? I need a life :(
Replicel minor update:
https://www.replicel.com/news/replicel-launches-the-next-stage-of-a-research-project-with-the-university-of-british-columbia-to-build-world-class-hair-follicle-cell-data-map
Pfff replicel didnt say anything new ! Whats going on with stemson therapeutic human trials that are set to begin this year ? Please thank you!
Researchers have found a way to heal skin tissue without scar formation… at least in mice :D
The crazy part is that those mice also generated new hair follicles in this process, might be interesting if this works for human heads as well after hair transplants! And the best thing is the drug has already been approved almost 20yrs ago by the FDA for a certain eye disease.
The drug is called Verteporfin.
Are any hair transplant surgeons using this yet to eliminate fue scarring?
That s sound crazy good no ? I mean if this could work, it would be the end of hairloss or almost for women in my case no ?
With 50 mg of spironolactone i don’t have hairloss…if this could regenerate hair follicles
then i would maintain my new hair follicle while on spiro…
But does that mean we would have to really damage our scalp to do so ?
I have tried taking spironolactone but felt it was messing up muscles. Did you have any sides ?
Hey Admin any thoughts? https://www.reuters.com/world/china/chinese-firms-covid-19-drug-claims-draw-scientists-skepticism-2021-05-14/
Wait what? Stemson doing human trials this year???
Gatdam if that’s true that was fast af
Stemson is our cure.. Unlimited supply of hair, for all forms of hair loss.. Forget all other bs, like Tsuji, or Histogen.
Stemson is it.
Tsuji said they will charge 1/4 million lol. Who’s gonna go to them??!
That’s IF they make it and bring the cure to market. And believe me some people out there are ready to sell their kidneys for the cure.
Its a cure, not a treatment. Every rich balding person in the world will get it.
You are already going down a path again where people will undoubtedly be disappointed in the end. If you read their statement in depth it clearly states, that they have fda clearance to enter a clinical trial for the condition “endometriosis”. This will not be a trial for male pattern baldness. In fact the term “hair” is only mentioned three times in that text and two times its part of the word “chairman” lol. I think they are going ahead with endometriosis for now because it would be a condition covered by insurance companies and the anti body will be very costly. I am afraid, that trials and development for AA will take much longer than that if they will happen at all. Thats the reality, hair loss is a non priority for companies like this.
https://markets.businessinsider.com/news/stocks/hope-medicine-inc-announced-dr-henri-nico-doods-as-president-and-head-of-research-clinical-development-1029887997
“Will enter phase II trials in multiple indications in 2021.”
Good chance that hair loss will ultimately not make it, but for now, fingers crossed.
Janey, stop spamming all forums with your personal opinions and don’t make them your reality. Thank you.
Who is Janey?
Hope medicine is working on a cream that I’m not going to be rubbing on my head.
@Marc: probably not going to be a cream, most likely genetic engineering with CRISPR CAS9.
Has anyone here used topical dutasteride from Strut? At $69 for a 30 day supply, I’m considering it, if it works well. I’d appreciate any feedback if there are any Strut users here.
@Rick – Have you looked into Happy Head’s topical formulation? They use fin instead of dut, but also higher concentration of minox. I might try that next year, but in the meantime, I’m experimenting with something else and want to give it 4-6 months. I think we’re all just biding time until something like Kintor comes out. Let’s face it – the Big 3 are less than ideal.
I use the topical finasteride from Strut for over a yr. Nothing positive to report. Good luck with the dut.
So how will the potent monoclonal prolactin (PRL) receptor antibody be administered? Like regerons antibody cocktail through an IV?
Hey, has anyone ever tried microneedling by itself (w/o minox, fina, etc.)? Only my temples are shedding from now, I’d like to try but not sure about the efficacy. Thx.
Dr. Chen answered everyone’s questions in the FAK inhibitor post FYI. Doubt he will have time to come back again, and the research is still not in human trials. So no use getting too excited this early in the game!
Admin –
Have a thought for you – and I have a fairly strong conviction that you’ll want to explore this path, If you feel secure it it’s outcome. Have you researched DAO’s, and how they democratize fundraising, etc? Most notably, a DAO backed my millions of individuals nearly purchased a copy of the constitution? Would love to chat with you about it if you’d like. I think this community could be converted… and be a contributor to the growth of a, or multiple, solutions. Those who would participate would be given first access to information and potentially trials – all within the boundaries and rules written within the DAO. Engagement would be super high…. And the publicity ……..(good for the industry)…. Worth a discussion piece, or maybe not. I’m glad to help but I think you should be the one running it – can explain why also. Well, let me know!
Hey Jon,,
Thanks for the idea! Unfortunately, I am starting a new contract job soon and also have to run this site and the Discord. Not yet sure I have time, but maybe you can e-mail me (see the contact us page for e-mail address) and I can star the e-mail for now?
Can you post in better quality please?
Unfortunately, the image size was too large and this was the best I could do after optimizing. Make sure you clicked on the image to open in a separate tab (where it looks a bit clearer and larger).
FYI — a reader set up the below for a better quality image:
https://www.zupimages.net/up/21/47/7332.png
I’m a chinese. This interview does’t mentioned anything about hairloss treatment and medicine.
Thank you :-(
Admin I can’t see anything taking n5 to n0 besides some kind of cloning dont you agree?
I don’t know much about Hope but I find the initiation of their Phase 2 trial very exciting. As I understand it, this is not just a maintenance treatment but potentially a hair growth treatment, given that the study in stump-tailed macaques, resulted in nearly a doubling of terminal hairs……………of course we all know how success in animals doesn’t translate well to humans. Still, I am hopeful that they are geared up for enrollment giving us the possibility of pictorial proof of success sometime in the next 12 months.
Sounds good, when will it appear on https://clinicaltrials.gov ???
I can see: https://clinicaltrials.gov/ct2/show/NCT05101317 for “Endometriosis-Associated Pain”
Thanks for checking Ollie. I will post in our Discord too in case some people feel like checking for updates regularly.
Admin would you categorize their approach as a potential cure for baldness or rather a more efficient treatment? I read somewhere their treatment includes minoxidil which does not make me optimistic tbh.
@ red its a treatment you have to rub a product or take meds or both at the same time ,it doesn’t sound as good as hair cloning.
Its injections, not some topical.
SAMiRNA and siRNAgen Therapeutics:
https://www.nature.com/articles/s41598-022-05544-w
HLT has good discussion:
https://www.hairlosstalk.com/interact/threads/published-in-nature-weekly-treatment-with-samirna-targeting-the-androgen-receptor-ameliorates-androgenetic-alopecia.136158/
These results suck and show no improvement. Why even post this. Its 2022 I thought we all should be more alert after all the scams the past 20 yrs. Sorry for coming on so strong but I’m tired of these companies showing junk and thinking they have a good treatment. Show me legit regrowth of at least a full Norwood then it will catch my attention.
Lately, lots of natural products have been popping up like mushrooms after the rain, this is not a good sign …
Lorence,
Well stated mate. I reckon that the focus should be on the big 3: HOPEMED, STEMSOM, MOOGENE
2022…crickets.
3022…Rogaine changes branding on its tin.
4022…hair loss finally cured.
4023…asteroid hits planet.
Summy its sad to see how your optimism and vision have decayed over the years. I remember you when you used to be so open to all these new treatments in research back in 2013. God bless and Godspeed.
Yes Aaron, in regards to hair loss, I cannot deny…my hope has been tested of late-that is true. Call me a disappointed optimist if you will. What scares me more is that I’ve been on here almost ten years! Cheers mate.
https://www.businesswire.com/news/home/20220201005458/en/Stemson-Therapeutics-Announces-Additions-To-Its-Scientific-Advisory-Board Stemson update, they’ve recruited a Dr who is using the same method (iPSCs) to retina cells using scaffold. This doctor has already started human clinical trials and is assisting Stemson’s team…..
It looks like this decade spells the last one for balding.
Let’s not exaggerate and keep our feet on the ground…
I’m mean, the pipeline is pretty promising but I will get excited when some of these products come to the drugstore counter tbh!
We are happy to announce that the manuscript entitled “Dual-Mode Vasodilator M119 Delivery to Hair Follicle via Dissolving Microneedle for Advanced Alopecia Treatment” was accepted for publication in Advanced Therapeutics.
Here, it is suggested that TOP-M119 (M119), a novel drug acting as a vasodilator in the scalp, and M119-loaded DMN with a specially designed shape can be used for the alopecia treatment.
https://onlinelibrary.wiley.com/doi/10.1002/adtp.202200052
Interesting find Mr. Bekoo.
Unfortunate News- cosmerna shut down their cosmetics it was rejected by the Eu just like Korea now they are gonna attempt clinical trials so 5-10 years we will know . Next in the group buy I’m in we have tried kx826 99% purity for over 3 months and we so heart side effects but no improvement or maintenance .
Dude Pegasus stfu bro we don’t need that negativity that they failed just use fin or dut and in 5 years we will have something.
what will we have in 5 years ? I am desperate. I am a female and 41. I be had maintenance through 20 years thank to the pill and 25 mg Spiro. Now i have to give up the pill because apparently at 40 you should not take estrogens anymore…i am desperate progestative only pill won’t work, and that no men will look at me, while now they all think i am this thing from heaven (clearly they re blind but i take it).. i feel my life is over, that i’ve been living as a fraud till now and that i am about to be discovered…i feel like shit.
You have no idea what this hairloss hell means for a woman, it is a death sentence.
Very sorry to hear you feel this way. I do understand you more than you could ever believe. But don’t do that mistake of thinking that hair define you, thinking that is a death sentence more than losing hair itself.
As a woman it does define you. In the media you see short women, tall women, skinny women, fat women, dwarves, women bald because they have cancer, sometimes women with hairloss because they have alopecia aerata or universalist (the clean kinds and acceptable kind)…never will you see a woman with androgenic alopecia.
If i meet a guy now, what should I do “a by the way, in a year from now i am might be as bald as you best buddy Robert, will you still be willing to see me naked and touch me ?” Seriously… I am a monster of nature, it is a sign to tell dudes to stay away from me… I am not able to deal with this.
Everyone’s body is biochemically unique. Different processes progress at unique (to you) rates. Why is the cutoff age for estrogens 40 instead of 39 or 41? Is it just a rule of thumb and not unique to you? Most women at 40 shouldn’t take estrogens? Or has a doctor told you something about you based on symptoms that you’re getting from taking estrogen?
Very interesting gene therapy for a disease (via blood stem cell modification) that also darkens hair:
https://www.theatlantic.com/science/archive/2022/06/experimental-gene-therapy-cystinosis-hair-color/661348/
Fukuda has been pretty active in terms of research – just this June:
https://www.researchgate.net/publication/361355988_Bioprinting_of_hair_follicle_germs_for_hair_regenerative_medicine
https://www.researchgate.net/publication/361406536_Reprogramming_of_three-dimensional_microenvironments_for_in_vitro_hair_follicle_induction
Seems that he is totally ready for his human trials. Unfortunately there is no information or website from his new company available. Maybe Admin you could ask him directly?
Thanks Ben. I updated and republished the Fukuda post and also added a new interesting video that you will like. I also e-mailed Dr. Fukuda. He did not respond the last time I tried, but fingers crossed.
HopefullyNot…have you tried rogaine? There are compound minoxidil that have finasteride in it that can work well topically for you to grow back hair. Also look into using essential oils like peppermint rosemary and aloe vera as well. This can help slow down loss. Try taking a hair vitamin and collagen. I heard the spirolactone works well for women. Even transplants work very well for women. Check out Dr Konior in Chicago. He deals with female pattern hair loss. If you have a more norwood pattern like a man instead of a woman lugwid…then you will hsve even more donor to use for more hair follicle. I’ve seen women almost bald like a dude get a fue transplant and got good coverage. Plus women can style their hair more than guys and cover any loss. Check out the stuff I listed. Hopefully this helps….just know many women lose their hair. I am noticing more lately. Eat healthy diet as well. Check your ferritin levels too. You could have low ferritin even if your iron levels show normal range. Demand your doctor check it along with your thyroid. Iron ferritin should be around 70 for optimal hair growth.
I am Spiro already my hairloss has been stable for almost 20 years. But i be to get off the combined pill and Spiro might not be enough, plus I cannot up my dosage because it upset my stomach…. I tried Rogaine, it never did anything for me. I take hair vitamins everyday, besides making my hair grow faster it does not do much. But thanks. As for hair transplant, i have a diffusé thinning and considered not a good candidate so far…. But thank you
I still have donor hair though, since I haven lost any hair at the back of my head, it is diffuse on the top and a bit on the side..people don’t notice so far cause my hair is short but i am afraid of getting off the pill
What are the type of minoxidil that contains finasteride ? Can I find them online ? I live in Europe… Thanks a lot.
Oral minoxidil could be an option too, just be aware of potential hypertrichosis. Dr Donovan has a good series of articles on it.
Best wishes.
Well, i am already on Spiro..i fear oral minox will give me side effects .i have mild side effects from Spiro..classic minox never did anything for me…maybe if it has finasteride in it…
Injections in the stomach? That’s interesting…that means systemic and not local delivery. That might also be the reason for relatively lengthy trial duration.
I saw that posting when it was initially published and was very impressed with his report, but didn’t take it all that serious. It has become very hard for me to believe anonymous experiences shared online.
Anyhow, I try to believe it and it’s good news. This could be a real game-changer. Bayer is a pharma-behemoth, and the monkey-trial results were unbelievably good and basically a cure.
Let’s keep an eye on HopeMed!
That guy has a 10 year history on Reddit and seems very legit. He mentions living in Australia in his past comments too. To see all his past (and future comments), here is the link:
https://www.reddit.com/user/moeman32/
Yes seems legit, I agree. I didn’t check his posting history. „Custom Lego statue designer“ is a profession I haven’t heard before…sounds like more fun than my job though ;-)
There also was a picture of his scalp (no face) online somewhere – it apparently was him but seems to have been removed now.
If (big if!) the pictured scalp was his, and I compare it to the dude‘s pictures in his profile than the result is nothing short of miraculous.
New update from the guy:
https://www.reddit.com/r/tressless/comments/ywii1s/bayer_medical_trial_follow_up_dose_4/
Saw that! Very encouraging.
Unfortunately, this will be very expensive (maybe 150K$ for 1 procedure, to be done every four year), and moreover this is years away :( I’m more interested in Kintor and its GT20029 cream, but this is years away too.
Ridiculous statement. It’s years away yet you know the price?
Jesus, give me a break. Enough internet for today.
Would a reduction in costs be viable with technological evolution? I would pay up to €50/60k if it were a definitive cure or in my 30s to 50s
They can sell it for 5k for 6 months but i don’t think they will. mAB for cosmetic purpose should cost around 15-20 (but that too is a lot for average people). Gipsy is quoting the price of antibodies used to treat cancer. And THIS IS NOT A CURE, you will have to take another 6month course after 3/5 years if you are not on AA.
Theres never been any mention of pricing on this so not sure where you got 150k from. Also the gentlemen in the tressless subreddit who is undergoing the treatment stated its a one time, 6 month treatment schedule of a certain amount if injects and the regrowth is believed to be permanent. Kintor is nothing compared to this. A topical anti androgen will do nothing for those already bald but this holds strong hope for excellent regrowth. This guy even stated that he has grown back 15 mm in hairline so far. You think kintor can do that? Not even close. This is game changing.
The results isn’t permanent.
Outstanding! Love the $56 million raised, coupled with leaks of product success.
Admin, is this currently the most promising cure, in your opinion?
Hard to discern from just one (albeit legit) Reddit testimonial at present, but encouraging for sure.
When people say “cure“ they always mean to bring back their hairline from NW7 to NW1.
But for all early hair loss suffers this is basically cure on long term. Just imagine if you can combine Hope Medicine and Kintor and go on every 5 to 7 years to repeat the treatment. It will be expensive, but you will stop your early stage hair loss and you will have full head of hairs.
Stem cells/iPSCs therapies and bioprinted hair follicles in 3D/4D culture are very far away from clinical use. Maybe 15 years from now. Mostly, because of safety reasons and highly cost to isolate and culture stem cells on large scale with high precision and quality.
@Bryan 100% spot on! I agree!
I haven’t had a chance to read all of the details and it is only the account of 1 person but such a quick response is absolutely incredible. I believe I read in the post where even the docs were in disbelief. Since the phase 2 trial of HMI-115 for women with endometriosis is already under way in the US, and since this is injected into the abdomen and not topically injected, I am wondering if we will hear/see about any anecdotal hair growth responses from women in the study. I guess it’s also worth pointing out that since the endometriosis trial has about a year head start, if it’s approved, no question it will go off label.
I am surprised that it’s only tested in USA and Australia? It’s from a Chinese company. Are there any trials in China? These Trials would be faster. Also Trials in Europe would be interesting because the Bayer company from Germany is involved, one of the biggest pharmacy companies in the world with 50 billion turnover and 100.000 employees.
Its is confusing, because Bayer was originally involved, but now it is all Hope Medicine. Also note that the monkey trials were in China.
Confusing things even more, there are concurrent trials for both endometriosis and AGA.
I read this on HLT: How did we get to this point? The antibody was developed by Bioinvent, and licensed to Bayer in 2008. Some years later Bayer finally got around to developing the drug for endometriosis. They conducted a phase I trial that was published in 2018. During the preclinical work for this trial it was noticed that the drug increased hair growth in mice. Bayer, however, was not interested in pursuing a drug for male pattern baldness. They had begun an initiative to focus on women’s health issues. Fortunately for us Rui-Ping Xiao was already working with Bayer on another project as head of the IMM primate center in Beijing. She asked Bayer if she could test the drug on bald, stumptailed macaques, and they agreed. When the results came in she formed Hopemed Inc., and purchased the global rights to the drug from Bayer in 2019.
Fascinating and disappinting at the same time! Same goes for delay in oral Minoxidil. Maybe even RISUG, if ultimately superior to Finasteride and Dutasteride.
https://www.hairlosscure2020.com/risug-male-contraceptive/
Admin, This article about halfway down summarizes how HMI-115 went from Bioinvent to Bayer to Hope. https://www.biostock.se/en/2022/05/bioinvent-shifts-into-higher-gear/
One article I read said this: “Its Phase II clinical trial for the treatment of androgenetic alopecia is an international multi-center, randomized, double-blind, placebo-controlled study, which is planned to be carried out in the United States, Australia and other countries.” So there may well be trials in Europe.
Are the phase 2 results from the US still expected in early 2023?
Never mind, I think it was Kintor results that were due then! So I guess my question is – when do we expect Phase II results from Hope Medicine?
The US clinical trial site was last updated in April of 2022, and at that time they had not yet begun recruiting. I don’t understand why you would start your trial in a country that requires a phase 1 trial (Australia) when you could start immediately in the US at phase 2. I would also note that currently they only list Victoria, Australia as a clinical trial site. https://clinicaltrials.gov/ct2/show/NCT05324293.
Dr. Sinclair seems to carry a lot of weight with Hope given the first trial to begin is in Australia and under inclusion criteria they are using “female subjects who meet Sinclair scales 2-4″…..as opposed to male subjects where they are using the Norwood scale.
I would also note that on Reddit, Moeman says that: “They can’t start phase 2 until the Aussie phase 1 is done or it can bias results. I am repeating what I was told by the researcher, who should know? Idk z:)” If true that would mean the US trial wouldn’t start until sometime mid year 2023. That doesn’t make a lot of sense to me but it did prompt me to take a closer look at the US trial and it is not a placebo controlled trial. It is “Open Label” which means both the evaluator and the trial subjects are aware of the drug they are getting. So everyone gets HMI-115. I encouragingly take that as a sign they are expecting clear undeniable results.
Pinotq, you are a very valuable source of info, thank you!
For sure!
Thanks Yoda and Admin………I think it’s called desperation :)! As you can guess, I am very excited about this possibility. I am reaching out to my doctor to see if he knows anything more from the insiders perspective. I’ll let you know if I hear anything new.
I’d like to be in that study. Any idea where they will be conducting the US trials?
That’s almost like the control-group in trials that test psychoactive substances like psilocybin or MDMA – it’s very obvious for patient and doctor who‘s given the the real medicine, because they trip hard and the others are without any reaction. So eventually they skipped the placebo, it made no sense.
Another special situation is when a investigational new drug is obviously so effective and safe that trials are being stopped – for ethic reasons. An example of this was „Paxlovid“, but I assume this rule only applies for life-saving drugs.
As things stand, HMI is also hugely effective and safe.
This is great news. Thank you for this update, Admin.
Also, for more inspiration, watch some spectator videos of the Artemis 1 launch. :-)
Holy moly! Combine the reddit posts with the monkey pics and this sounds pretty legit thus far! So much so that I’ll gladly take on the role of Nasa_rs as it relates to this. This is to me, what Jak inhibitors were to him! Cure is coming… Christmas 2026! lol! Let’s go!!! ;)
In all seriousness, I’ll be following this closely, and remaining cautiously optimistic. Great update! Thanks, admin!
PS, I can’t help but laugh that the name of this blog has become comparable to “in the year 2000” on Conan O’brien. It would have been great to have hit a “homerun” by 2020 (or really… “in the year 2000”), but there’s progress being made. Seeing money flowing in for research and clinical trials gives me hope. Worst case scenario, even if this fizzles out they will have learned something that can be applied to the next thing.
I’m pretty darn excited. I’ll always be a little skeptical (of everything), which I think is healthy in this arena, but it looks and sounds like the real deal. Man, I really hope this makes it all the way through and continues to impress. The results so far are amazing.
Merry Christmas Admin, thanks for your informative posts for another year!
Does anyone know if there are or there will be trials in EU? It would be really disappointing if EU was late on this one.
@Admin
Thanks for the update.
How many doses he took and how many doses he will take until the end of trials?
It is a little confusing as he said two different things. His latest update is titled as “midway point”, so 16 doses total (8 remaining)?. But one of his later comments says “Its a 6 month treatment plan. 12 injections over 24 weeks.”
So, he has 4 more or 8 more doses until till end of the trials.
It is big difference if he has 4 more left than 8 injections.
One thing that I come in mind was his replay that “this will be one time treatment“, so he believe that results will last very long time.
I think they skipped a dose for some reason and got one early? Probably he’s confused. Supposed to be 13 doses over 24 weeks – one dose every two weeks.
I know this is years away but this guy is a legend for all these updates. Really seems to be working and he’s been bald for 17 years. And really, who knows. Maybe it would be fast tracked in another country (those flying in every two weeks for an injection isn’t really feasible for most).
This is still at least 8 years away from commercialization.
We are probably the last bald generation.
The commercialisation date for the same drug for the treatment of endometriosis is planned for 2026/27, it’s unlikely that the commercialisation for hair will take much longer, I know they throw around 10 years on reddit, but that’s just an estimate for Austraila which is very strict. If the drug proves itself to be very safe there are always options for fast tracking, also it’s greenlit for phase 2 in other countries already so in the end nobody can tell how long it will take, so I wouldn’t focus so much on random numbers on reddit.
Same with the supposed price because nobody can know that now
Don’t reply to the known trolls.
Bryan wants to spread his agenda including pessimism and bad vibes.
I still have the suspicion that the Australian trial is part of phase 2, but probably not for Australia itself (stricter laws I heard). Phase 1 is done and sealed since 2020. It’s confusing. One indication for this theory: they are using the final amount of antibodies like in the monkey trial. It’s not about toxicology or dosing – which a trial 1 always is about – it’s clearly about efficacy.
Telling someone the truth doesn’t mean that someone is trolling and spreading pessimism.
Thanks for the heads up!
I just felt like clarifying, since people on reddit are also panicking about this as well…
According to the OP on reddit it is a phase 1 trial (eventhough now that you mention it an “odd” one), since Hope initially wanted to use the results of the Endometriosis trial data for the phase 1 trial but Australian government denied it, so the current trial is now the phase 1 (he also said that in Australia they need a phase 4 for it).
But I of course don’t know how correct the info is .
Also, doesn’t the pipeline page on their website indicate it’s in PhII?
As a woman, would it be reasonable to ask for this even without endometriosis if available in 2026/2027 ?
It’s all a guess on commercialization. They don’t even know. There are too many variables. But I wouldn’t guess 8 years away. Maybe 4-5, but of course, like I said, nobody knows for sure and there are always unforeseen roadblocks and obstacles. Always. It very well could be eight, but I don’t think so. Not if it’s proven to work and proven to be safe.
I’m excited that age doesn’t seem to factor in much, nor does the amour of hair loss. The Reddit guy can’t be too young – he said he’s been bald for 17 years. And it’s working for him. Time will tell how well it really works. He’s halfway. I can’t wait for actual human photos. I remain skeptical but hopeful. The Reddit guy can’t post photos but in his explanation it sounds like it’s growing hair even in totally bald areas. Exciting stuff.
Thanks, Admin. Appreciate all the updates.
Guys who are now 10 or 12 years old until their middle twenties they will not give a care about hair loss, because they will have at least 4-5 very effective new hair loss treatments + fin/min/hair transpalnt. They can stop hair loss at very early stage, get good regrowth and maintain their norwood on very long term.
Even there are some new treatments in next 8 years they will be very expensive, repetetive and for tiny minority of the population.
I put too much hope on hair multiplication/ hair clonning, but that is at least 20 years for ordinary Joe to get it on his head.
Yes, there are more and more scientific papers about iPSCs/bioengineerd hair follicles or organoids, but they are still on papers for scientific purpose. Nothing more. Bringing scientific paper to actual product is very long period, lot of money, lot of dedication, lot of work.
I’m surprised there’s not more skepticism about this Reddit guy. How do we know he’s real?
That’s a valid question. Who on earth knows 100%? Let’s hope he’s legit.
I agree with you guys, hopefully the Reddit guy is legit but trolls troll for a whole host of reasons. Not least of which they derive some sort of twisted pleasure from it.
Is it common for people in trials to come on Reddit like this? I don’t remember seeing it and I’m not sure I’d do it. However I actually do see the appeal to claiming miraculous results to what will at some point be millions of people if he keeps going.
The science seems promising but I can’t bring myself to put any value in the Reddit posts personally.
A large part of my trust is based on the fact that this guy has been on Reddit for over 10 years, and has a number of past posts about Australia. The Sinclair clinic is also way too renowned in the hair loss world to try any kind of gimmicks such as this in my opinion.
Yeah that’s definitely a good sign.
I’m not critical of anyone advertising the research. It has to be done and the commentary in this post is great. Just wanted to know anyone’s opinions on the guy…his account does seem like a real account so that’s good.
I believe he shared pics of his trial paperwork with some skeptical users, who then vouched for him.
Admin; as a former patient of Sinclair Dermatology, I would err on the side of caution. Not wanting to pop anyone’s bubble, and I have no doubt the trial is legit and Sinclair has a big rep, but in my experience the doc’s there rush you in and out quickly and are a bit unprofessional in their manner. I hope this doesn’t translate to their trials?
I’ve seen two docs there. One was nice, but quite slap-dash in her manner. She quickly took my pulse and concluded I wouldn’t have any problems with Min. At one stage she suddenly pulled out her camera and started snapping ‘scalpies’ (in terrible light I might add). I know from experience your head must be locked in place with the same exact angles and flash lighting – including hair parting, etc. But, whatever – it seemed rushed.
The second doc was a phone consult a few years later. She was curt and dismissive when it came to any questions I had. Given my own health (including the hefty consultation fee), I don’t think it’s unreasonable to ask questions about anything I’m prescribed or any expectations I may have. I basically couldn’t get a word in.
Several friends of mine have gone there for different medical reasons and experienced varying degrees of satisfaction also. Some good. Some not so.
It’s a well known derm in Melbourne so it’s a very busy place, but the reception area felt like a cattle yard. Apart from their salaries, I don’t envy the docs working there.
With AGA hair loss, they put you on a 3 monthly (AUD $300) check up cycle which ends up being very expensive. Their compounded Fin/Min caps or oral Min caps on the other hand are reasonably priced, but you can get the same thing at a good compounding pharmacy without the endless ‘consultations’ and much cheaper to boot. To be fair, they obviously want to monitor your early progress and any complications that may arise, but after nine months or so – I don’t see the point? (Other than make more money perhaps.)
Their ‘Hairy Pill’ TV ads too were pretty tacky to say the least. It wasn’t a good look. Even the second doc I spoke to from Sinclair (who shall remain nameless) dismissed the pills saying they’re just Fin/Min with some vitamins added and not as good as their in-house compounding!!!! Go figure.
You can take this all with a big pinch-of-salt of coarse, but my previous local doc said Dr Rod is widely known as being very arrogant resulting in him not referring his patients to Sinclair anymore (after their complaints about his demeanour).
Also, I was quite shocked to hear a derm warned a friend of mine, who was initially referred by his doc to Dr Rod for a scalp skin cancer removal, to steer well clear as his surgery skills are considered well under par. Professional jealousy maybe? Who knows? All just hearsay I know, but worth noting when it comes to the current trial.
I understand some other patients of Sinclair Derm might be contrary to this, and that’s fine, but I can only go on my own experiences with them – which wasn’t 100% positive in terms of their professionalism.
To their credit however, they did conduct a PRP trial years ago and concluded it was ineffectual, (which I experienced also) including extensive research and trials into oral Min – so they do have some cred for sure, but I would just wait an see how it all pans out. I hope the “Reddit Guy” is real and HMI-115 goes well for him and he doesn’t end up with a head like a Jackfruit after all the injections. I’ll be interested in the Phase 2 results in the States when they happen.
Thank you Summy, this is great info. I hope this volunteer gets very-obvious-to-his-eyes regrowth rather than relying on the clinic’s data.
Thanks for the thoughtful post Summy, it’s real world experiences like this that are the most valuable info! Agree that tempering expectations is the best course as we’ve been down this road many times. Fingers crossed, hoping for the best. Happy 2023 everyone!
Happy to help fellas. Happy New Year.
Sorry to post again on this subject but I’m trying to understand the science here.
This research is about degrading the prolactin receptor correct? I believe Prolactin itself won’t be affected by this.
I’m curious because there are medicines that have side effects of increasing prolactin or reducing it.
There’s nothing to indicate that reducing prolactin could be helpful for hair loss? Similarly I assume increasing prolactin would have permanent affects on hair loss?
I’m trying to understand the science here.
This research is about degrading the prolactin receptor correct? I believe Prolactin itself won’t be affected bu this.
I’m curious because there are medicines that have side effects of increasing prolactin or reducing it.
There’s nothing to indicate that reducing prolactin could be helpful for hair loss? Similarly I assume increasing prolactin would have permanent affects on hair loss?
Hi can you stick to one username if possible? You also posted same comment under “JoeJohn”.
My fault. The post wouldn’t go through so I changed the name and tried again. I guess they both sent. You can delete either one, that was an accident.
No worries. Sometimes I also take a half day to approve when busy, or they automatically go to spam.
As the two medicine are the same, if endometriosis come out first, can I prescribe it to treat the hair loss?
That’s an interesting take. I think the Emdometriosis trial is at least one year ahead.
If it’s exactly the same drug, then there will be definitely lots of men with endometriosis ;-)
Off-label-use is not unusual.
Yes, it just like proscar intended to treating prostate, but we buy it for hairliss.
Not sure as the hairloss treatment is through local injection, and not as a pill you take everyday..
I’m just read that “The application of PRLR blocking antibodies inhibited endometriosis development to the same extent as anti-estrogenic drugs.”
= So does that mean that it will also counteract the effect of estrogens on hair ? I find it weird cause estradiol is important for hair…
The treatment should be through injection, and I thing they are the same medicine. The problem is how to convince the doctor to prescribe the medicine to men which do not have endometriosis problem
If the endometriosis drug is the same, works for hair loss and can be administered properly then there are docs that would prescribe it for hair loss. Maybe not a lot docs until it’s approved. This is how I originally accessed topical minox back in the mid-80’s and Proscar (Propecia) in the mid-90’s before either were approved for hair loss.
Avodart too, till today :-) Except in Japan and South Korea.
Good point Admin. Docs can prescribe and administer pretty much any approved drug for off-label use. However, all the hair loss drugs mentioned weren’t cost prohibitive to “pay out of pocket”, as insurance won’t cover off-label use. It’s possible that the endometriosis drug will be covered by insurance and could cost more than the hair loss version, which wouldn’t be covered by insurance (unfortunately).
Is there any grey channel that can get the HMI-115 after they finish phase 2 trial.
This is probably the one I’m most excited about. I’m early in the norwoods but I have a level of diffusion that when I get into my mid thirties, meds or no, will start being readily apparent. This is an absolute game changer if proven efficacious not only for my selfish desires but for those who long ago gave up or couldnt tolerate treatments now being able to maybe have a shot! The monkey studies were honestly great especially since it was on stump tailed macaques and not rats like most (I’ve grown to hate rat studies over the years).
Is your diffuse thinning in the donor area? You’re smart to realize the diffuse thinning would be helpful at first and bad in the end. That’s what happened to me. I read that the prolactin might be particular relevant in female pattern hair loss which is basically diffuse thinners so perhaps this treatment would have unique importance to your case.
What happened to SCUBE3?
@lorence If i remember correctly scube3 is still in preclinical they need couple years to start clinical trials. Amplifica ->the company behind this molecule should start clinical trials with some different molecules other than scube3 at the first quarter of 2023 so its a little dissapointing for the hairloss veterans overally.
Yes, if a giant like Bayer is involved … this might be it.
Bogdan, there‘s only one reason that SCUBE3 is „on pause“: it‘s an
endogenous protein and hence not patentable and therefore unmarketable (or difficult at least).
Amplifica is working on other proprietory molecules which affect SCUBE3.
We really should be looking into SCUBE3 (buyable already!) ourselves. Amplifica won‘t do it…
Still think they are a great company though.
Just can’t wait until it got final approval….
Let’s figure out how to get the HMI-115 in some special channel.
But I’m excited about this drug hmi-115. This is really super promissing in terms of efficacy. The problem is that years ago i was exited about Histogen and we all know how it ended:D
I’m curious about HMI-115 too, but we’ve been here before many times it seems. If one of these players actually makes it out of the gates, then great, we can all dance in the streets and look forward to having potential hair like Java Man. Otherwise, my advice would be to temper any expectations at this point…then you won’t be too disappointed if it happens to bomb (like Histo did).
It’s possible he’s trolling but I think he’s legit. I have no proof of course but I think it’s true. Either way, there are no photos from the study and until there are I won’t get my hopes up too far. Some people think “good growth” is ten hairs. I need photos. I do like his updates though and if i was a betting man I’d say he’s legit.
How proprietary is this injection? In one of the early posts from 3 years ago a commentator said it could be replicated by a lab given info Bayer published. If it gets through phase 2 with promising results is there any chance some shady clinic finds a way to offer this?
Would you trust a shady clinic giving you 24 weeks worth of shots? I know I wouldn’t. Mess around and find yourself with a third butt cheek (or eye I guess is a better example, but not nearly as funny). Nah man.
Just can’t wait until it got final approval….
Let’s figure out how to get the HMI-115 in some special channel.
(Hi admin, please delete my previous comment reply to Ben, thanks.)
I found something interesting in the internet, it says that docs in California have the right to prescribe unapproved medicine to patients.
https://www.npr.org/sections/health-shots/2017/03/03/517796956/patients-demand-the-right-to-try-experimental-drugs-but-costs-can-be-steep
Another anecdotal report from someone whose friend is in the endometriosis trial
https://www.reddit.com/r/FemaleHairLoss/comments/104ieap/endometriosis_pain_relieving_trial_that_gives_a/
“My friend is going through this and she’s found a lot of relief. She also has AGA and has grown back a lot of her hair.”
I think the below is the clinic’s page on the Hope trial:
https://www.tidewaterclinresearch.com/endometriosis-1.shtml
Thank you, Admin!
I would think if this is a worthy wonder treatment, it will be purchased this year by big pharma. I got my fingers crossed. A larger pharmaceutical company would also help expedite the clinical testing.
Bayer is a huge pharmaceutical conglomerate.
Omg. You’re right Sam. Thank you. I forgot. I previously assumed they bought the patent from Bayer. I see that it is a license agreement. Big farma has kind of been on board all along.
Ok. From Pinotq November comments above, it appears HopeMed did eventually buy it from Bayer. I guess it doesn’t really matter. We should have an answer to the success of this drug sometime this year.
Latest update he says only 25% in the front (based on photos they showed him)? And it’s close to done? I’m not as impressed as I was a few weeks ago but I guess we’ll see what it looks like in the end.
The macaques only showed significant improvement at the 1 year mark, 6 months after the end of the injection. I would say the progress he’s reporting is very impressive. I just hope he keeps that subreddit updated with his progress after the end of the trial, hopefully posting before / after to see his progress.
It’s semi impressive. I mean, I’m not 100% bald so 25% would be great – esp if, like you said, it’s more by the one year mark. I’ll take it. No doubt about it (nothing currently on the market comes close to that). But if someone is 100% (shiny) bald, 25% (or even 35-40%) isn’t going to “cure” them. It would end up helping but not curing. But you’re right that it’s too early to say how this will turn out.
Pretty sure he said early on that they told him he can’t post photos until the trial is over…meaning he can (hopefully) at the end of this. Fingers crossed, I’d love to see them.
And 100 percent growth in vertex:
“The whole vortex space has filled out to twice as much.”
Yeah that has me curious. The front sounds meh but 100% vortex…but 100% increase of 10 hairs wouldn’t be good (ha devil is in the details) but you’re right he says it filled out. I wonder why all these things help with the back or top more than the front. Even minox. It’s odd. I’m sure there’s a scientific reason but it’s a bummer. Great to have hair in the back/top but if you’re forehead is a mile long it’s not ideal. But then maybe you could eventually use the vortex hair for a transplant?
I know, I know I’m hard to please lol
The HMI-115 posts have been deleted from the one guy posting results:
Bayer Trial dose 9. update
Sorry, this post was deleted by the person who originally posted it.
It doesn’t appear in any feeds, and anyone with a direct link to it will see a message like this one.
Ok. I just found another update from moeman32. He had to quit providing updates or he would lose his place in the trial.
He also appears to have become unhinged. He is really upset. After reading his latest comments he has lost merit with me. This whole thing is full of unnecessary drama. I’m also questioning if he truly got permission to post his results.
Yes, I added the update yesterday. Thanks.
Can someone update me on what happened here? The update in this thread said he showed pictures on dose 9? Did he really do that and was that when he bailed?
Afaik, he never shared photos publicly. He was clear on that point from the beginning. He bailed because someone reported him to one of the scientists behind the trial (for the lols). He was also upset because some people were threatening to commit suicide unless he gave them what they wanted (info/photos).
Why he deleted his posts:
https://www.reddit.com/r/tressless/comments/10wuc1n/why_i_had_to_delete_my_posts_about_any_trial_i/
https://youtu.be/Y9_1tR3WM0Y
I will understand if you do not want to post this guys material, but he seems to do a good amount of research on his hair loss studies.
This might help curb the HMI115 enthusiasm.
I don’t understand his point. He says dht decreases prolactin. So that would actually help explain why lowering dht is not enough since prolactin may increase when doing so.
We also don’t know how much prolactin affects hair. If its affect greater than other mechanisms of follicle destruction then lowering it may be more helpful.
Oh, Kevin, you so silly.
Once you sift through all the manure that’s typical for a Kevin Mann video, it turns out there are only two interesting claims he makes, and surprise: they’re also horseshit.
Claim 1: There’s apparently no way HMI-115 can work better than finasteride… except, to support this, he provides no actual evidence (an impossibly high bar, I know) and instead just throws “DHT is the master trash hormone” at us (how old is this guy?). Oh, also, HMI-115 does work better than finasteride in a primate model of androgenic alopecia. It’s right there in the patent for it.
I wonder if DHT is the master trash hormone for stump-tailed “mah cockies”, too, or if they have a different hormone they like to yell about on YouTube.
Claim 2: Prolactin suppression probably causes the same sexual side effects as DHT suppression… except low serum prolactin is only *correlated* with sexual dysfunction, and *causation* has not been shown (he even acknowledges this, so why is he wasting viewers’ time with this argument?). He also fails to mention that people systemically nuke their prolactin levels all the time for the purpose of *improving* their sexual function, and it works. Seriously. Look up cabergoline.
In sum, I can’t believe I made myself sit through 13 minutes of incoherent yelling for this. Congratulations, Kevin. Your arguments might not stand up to the slightest scrutiny, but you’re the real winner here.
I have to admit, he’s trying my patience and his trolling tendencies are really unpleasant.
Claim 1: There’s apparently no way HMI-115 can work better than finasteride… except, to support this, he provides no actual evidence (an impossibly high bar, I know) and instead just throws “DHT is the master trash hormone” at us (how old is this guy?). Oh, also, HMI-115 does work better than finasteride in a primate model of androgenic alopecia. It’s right there in the patent for it.
I wonder if DHT is the master trash hormone for stump-tailed “mah cockies”, too, or if they have a different hormone they like to yell about on YouTube.
Claim 2: Prolactin suppression probably causes the same sexual side effects as DHT suppression… except low serum prolactin is only *correlated* with sexual dysfunction, and *causation* has not been shown (he even acknowledges this, so why is he wasting viewers’ time with this argument?). He also fails to mention that people systemically nuke their prolactin levels all the time for the purpose of *improving* their sexual function, and it works. Seriously. Look up cabergoline.
In sum, I can’t believe I made myself sit through 13 minutes of incoherent yelling for this. Congratulations, Kevin. Your arguments might not stand up to the slightest scrutiny, but you’re the real winner here.
Hope medicine got a new CEO.
https://cnmobile.prnasia.com/story/392917-1.shtml
Also they got approval for clinical trials phase 2 in China
https://mp-weixin-qq-com.translate.goog/s/kPcPIH5_2LpBV7bkPn217A?_x_tr_sl=zh-CN&_x_tr_tl=en&_x_tr_hl=en&_x_tr_pto=wapp
Thanks for the update.
Not sure if this has been posted before. I just noticed on Hope Med’s website a little more info regarding the timeline. It says the Phase 1 in Australia ” is expected to be completed in the beginning of 2023.” My understanding is that injections for men have been completed so it appears this is relatively on schedule. Hoping for some pics by June/July at the latest. The website then says that the “phase 2 clinical trial is expected to be completed in 2024.” Interestingly, it also says that the phase 2 for endometriosis is “expected to be completed in 2024” …. even though the study started in October of 2021. So according to this, it doesn’t sound like approval of HMI-115 for endo could be obtained any earlier (and used off label) than the the approval for androgenic alopecia. It also says that Dr. Sinclair and Professor Jianzhong Zhang, director of Dermatology Department of Peking University People’s Hospital in China, will guide the planned phase 2 clinical trial.
They have revised the media article and the time frame (2024) has been deleted.
https://www.reddit.com/r/tressless/comments/13ty9n5/the_moment_you_have_waited_for_has_arrived/jm1k8ty/?context=3
Joke post by moeman32, but he made a serious comment.
HE SAYS IT WORKED BOYS
Yes, that’s very encouraging.
He also still seems to be loosing his mind. Doesn’t sound like a guy who experienced a miracle. When we heard his original claims we figured it would go down hill but you could never have expected this implosion. I hope he’s ok and I hope he has hair but this all seems ridiculous.
It’s working, see the picture reveal from Reddit:
https://www.reddit.com/r/Hairloss/comments/16ewgu2/hmi115_is_working_well_for_moeman/
Very clear privacy violation, but I can’t deny being a bit excited.
Had a consolatory feeling, after years suffering, finally see a promising drugs.
Recruitment for the phase 2 trial in China has already started in August:
https://mp.weixin.qq.com/s?__biz=MzIyMjU4Mjk3Ng==&mid=2247489130&idx=1&sn=6dc82aff61c878f798f2750afe237278&chksm=e82a17c5df5d9ed3518990e52561671aa0aeca8ac62d27cd6ac096d3b64a83ba51f7a36ea047&scene=27
Australian Phase 1 trials completed on October 12, 2023:
https://clinicaltrials.gov/study/NCT05324293
Apparently Chime Biologics and Hope Medicine had an agreement to speed up the launch. I guess this is a good sign even though we don’t have any recent data. I’ve seen also some photos and someone was claiming that they belonged to that guy who used to give info regarding trials and those photos were kinda disappointing. Hard to believe anything until they will reveal more info.
The photos (if really from this trial) were pretty promising actually, they showed about 30-40% regrowth in less then 6 months.
I always wonder what you guys expect in such a short amount of time, a haircycle takes ~3 months. Some people need to take finastaride up to a year to see results.
If you look at the monkeys, they also had the most regrowth between year 1-2.
Moeman also noted a few days ago that, since it’s phase 1 trial, they were given the same dose as the monkeys which means there big difference in size & weight, so an adjusted dosis for humans may yield even better results
https://www.prnewswire.com/news-releases/chime-biologics-and-hope-medicine-enter-manufacturing-agreement-to-speed-up-the-launch-of-first-in-class-antibody-drug-hmi-115-targeting-endometriosis-and-androgenic-alopecia-301968968.html
They are talking about phase 3 already?
The information regarding HopeMed is conflicting often times. It’s a private company, I think we have to get used to it.
Only the endometriosis trail is in phase 3 & there is even a trail for endometriosis in the EU currently. Its sometimes confusing which application is referred to since the whole pipeline started for endometriosis started earlier then AGA , Bayer originally disregarded the hair regrowth factor completely because the focus was only on endometriosis. Only after Hope bought it the AGA aspect was more looked into.
Thanks admin for covering this news. At first sight maybe not really worth mentioning.
But this collaboration is probably worth millions of investment from HopeMed. Which in turn means they must be convinced that this works.
Chime Biologics has 400 employees according to their website and is one of the leading CDMOs in China.
This is good news, lads.
Thanks Ben, yes this is great news. Two people here and one on Discord also mentioned. I am too tired and did not go through my entire post to clean up and write about my positive feeling.
Glad they started Phase 2 in China.
Need to find out when the (delayed?) US Phase 2 trials start. Also need to keep track of Australian Phase 1 results when they come.
Seems like Kintor and Hope Medicine are both on fire, and moving along at Chinese Communist Party dictated 5-year plan speed.
Hey Admin do you believe that Australia regulations will be move along faster compared to the US FDA trials?
In this case, Australia trials lagging China and the US it seems.
Did they even trial it in the USA for AA?
Of course it did, they got approved for phase 2. Scroll to the top and you’ll see
Admin this is a very exciting one for sure! Going into an agreement with Chime Biolgics to manufacture Hmi115 is a big deal, they just finished phase I recently in only June-July this year and they’re already making further moves, hopefully it will be successful… i think the race is on! between Kintor, Hopemed, and Dermaliq, and maybe even Tsuji to reach a cure or something much better than the big Three at least.
Mice have 10 hair loss cures.
Monkeys now even have a cure.
If they can now just do humans, we can all celebrate! It’s so close :-)
Admin, how do you see chinese treatments being approved by FDA/EMA when trials are done in China ? When I see Winveli for acne it is still not available in Europe 2 years after FDA approval in US.
So it took 4 years for that monkey to grow hair back from this treatment? Am I reading that correctly?
… c’mon man, read it again.
Treatment over the course of 6 months but after treatment it continues to grow hair up to 4 years I believe.
Looks like “moeman” is still replying in some threads. Have not tracked his comments recently after all the controversies and deletions.
https://www.reddit.com/r/tressless/comments/17i1w6t/hmi115_update_chime_biologics_and_hope_medicine/
Is it possible that Hope Medicine skip the Phase II Trial in EU and US and directly start a Phase III trial in EU an US in 2025 After Phase II Trial in China was finished? So Can Hope Medicine take the Phase II results of China to apply in FDA and EU to start Phase III there?
There’s no law that you have to undergo trials in the country where you want to market your product…as long as HopeMed shows the necessary transparency and does the paperwork AND most importantly fulfills FDA-safety-standards they should be fine.
By the way: trial number 2 is also registered at „clinicaltrials.gov“ which is a US-institution. This trial is solely held in China.
https://clinicaltrials.gov/study/NCT06118866?intr=Hmi-115&rank=3
PR Newswire Reports positive outcome established I phase 1b study of hmi 115
Moeman said something very interesting regarding dosage in that this was a safety trial and they were dosed the same as the monkeys and still had exceptional regrowth. I cant imagine how much better the hair regrowth would be if it was dosed for an adult human and not a macaque.
I have yet to find somebody (maybe here in the forum?) who can tell me if you have to adjust antibody-doses to bodyweight. I could find some information online but it was conflicting.
Most common antibody-treatments are standardized – if you weigh 45 kg or 160 kg, you get the same dose.
Macaques weigh approximately 10 kg, and the Australian trial for humans used the same dosage (120 mg per injection). But the China-trial only uses one higher dosed arm of intervention which is double that: 240 mg per injection. If you want to balance the weight-dose-ratio, you would have to inject around 1000 mg compared to the monkeys.
Maybe a more frequent injection-schedule can counteract a lower dose, but this also not tested in the China-trial (every fortnight).
It’s a little confusing to me, but who am I to question the experts.
Phase I is just a safety trial to make sure there aren’t any surprises such a severe immune reaction or other toxicity. Phase 2 is where dose is escalated to find max benefit.
Depends on the drug/antibody and mechanism. Once completed a Phase 2 if majority of patients respond well to one dose that could be the standard. Long term it may just not be in the financial interest of the company to narrow down a specific mg/kg of a particular antibody. Especially if you consider androgenetic alopecia where it’s older patients who are likely generally healthy. If it was a rare debilitaing disease in children you’d almost certainly be required to dial in optimal mg/kg.
It depends…. But the margin for weight in generous with most antibodies so there often is just a recommended base dose for adults.
I was treated with antibody injections in the past for longer period of time.
Most antibodies injections come on prepacked syringes already dosed. So there is little individual dosing involved.
There is also the matter involved that the body only processes a certain amount at once, so shooting over a certain dose is just a waste of material. That’s maybe why the cap it at 240mg for now…
That’s not really a convincing result in terms of numbers, but Moeman‘s pictures looked definitely better than these numbers indicate.
Good thing it’s obviously safe and that was the sole reason for this trial. And higher doses could be more effective too.
I’m fairly optimistic for this one, but I don’t think this will be the miracle we were hoping for. The monkey pictures are still insane though.
He was the best responder.
From the monkey images it’s visible that that the biggest improvement happened between 1-2 years while the study only lasted 6 months so who knows what happens in 1 year from now. I’m also not sure how much more frequent hair cycles are for macaques. I think we also need to give it a bit more time to write it off as unimpressive. I’m very interested what results the higher dose will yield…
I’m hopeful but let’s see how the second trial goes. And I want photos. Screw the numbers, I want to SEE the results. End of 24 is solid, if they can pull it off.
Nice.. That’s really positive news. Thank you for this update, Admin. Fingers crossed for more progress.. Cheers!
14 hairs per cm2 is garbage. If you look at other studies, that is well within the standard response for the placebo control group. The average person with hair has between 100-200 hairs per cm2. The whole reason the study with the monkeys was exciting is because they reported that in at least some cases they regrew 100 hairs per cm2. This study does NOT reveal anything impressive or interesting in regards to effectiveness.
If the average trial participant still had 50 hairs per cm2, then a new 14 hairs per cm2 is a good improvement.
Keep in mind that even after 2 years monkeys had regrowth, also their lifecycle is faster, I wonder if for humans it takes longer to see even better results. Also that guy who reported on reddit said that at the moment he is still experiencing hair growth so this treatment requires long term observations. I wonder if in the next trial the dose will be higher, maybe that will rush the hair growth.
The Reddit-guy (Moeman) just today confirmed again that he is still experiencing regrowth.
That’s remarkable as his last treatment was in April 2023.
Thanks Ben!
The results are not what I was hoping for and the lack of any pics calls into question how significant the 14 hairs on average really are but I think there is still much to learn. Even if it turns out to be more of a maintence treatment that would be a big step forward from my perspective given that I am not maintaining on Dut and OM. And HMI seems to have longer term staying power. I would really like to phase out my current routine and get rid of daily pills (from an overall health perspective) Plus, this is a totally new method of action giving hope that there will be at least some noticable regrowth. When I told a very accomplished doctor about HMI, he was stunned, and slightly embarrased, when I told him it didn’t act on the androgenn receptors but the prolcatin receptors.
This is what worried me after my hair transplant, being subject to daily medication for the rest of your life, I have been taking it for 4 years and I hope that news will soon arrive from Fukuda, whose solution is not a medication to prolong what is written genetically but to replace hair affected by alopecia with immune hair clones. The ideal would be an injection of cells that will regenerate hair in situ to avoid the trauma of an operation and the shock loss, but it seems that this will be impossible for years or impossible because there are hidden interests.
If they just test the safety with a low dose in phase 1, is it legal to increase the dose in phase 2 or later on?
Maybe the higher dose isn’t safe any more and they have to do a safety phase again?
They won’t increase the dose for phase 2. They also don’t increase the frequency. We already know that since August 2023 when they started recruitment for phase 2.
People need to stop coping and accept that this won’t cure you. Theres still a big chance that it won’t even make it to the market.
If it does get released in 5-10 years it will be an expensive maintenance at best.
It’s still better than nothing but everyone is just disappointed because the monkeys were basically cured.
Only six months results. Could grow hair up to two years. Why would you hire chime biologics to globaly market hmi-115 if it doesn’t work?
Good point David….now how do we get them to move this along faster? At this pace we will be waiting for results for a looong time.
Admin does a great job, and I think it was justified to have talked about the Reddit poster. But it’s delusional to say this does anything but completely invalidate the Reddit guy. His pics have seemed sketchy, his behavior seems clinical, and it now appears that he has executed one of the greatest reddit trolls of all time.
No one should apologize for him.
admin, just wanted to draw your attention to this, in case you hadn’t yet seen it.
https://onlinelibrary.wiley.com/doi/full/10.1111/jocd.16173
Thanks! Was about to publish a new post today, but will update my old fractional laser one first.
https://www.pharmnews.com/news/articleView.html?idxno=239361
New investment for HopeMed. They’re definitely planning for a phase 3…
New pictures from the Chinese trial. I hope these are legit. Looks pretty good for 2 months progress?
https://www.reddit.com/r/HMI_115/s/08DGbyvOM8
Someone sent me these a couple of weeks ago from a “leaked Discord” too… was not sure if legit.
Those pics must be legit, would anyone lie about results that modest?
I m relying heavily on amplifica. If this also produce underwhelming results. I will consider wearing wig with heavy heart. I am in a panic mode today. I am a woman by the way.
Because of my child bearing age and wanting for one. I could never try any treatment. N having difficulty in conceiving it.
Just waiting if and when I will have second child I will try minox N finasteride.
But the waiting has spanned for for more than six years. The damage is devastating.
Totally clueless people.
Stay Strong Gurvinder.
I’m a male and losing nearly 50+ hair from all sides(DUPA).
Never think about what other’s think about ourselves.