Yesterday, Aclaris announced that their oral ATI-501 JAK 1/3 inhibitor Phase 2 clinical trials were successful in treating patients with alopecia areata (AA). No serious side effects reported.
Not a huge surprise since JAK inhibitors have been proven to work well on alopecia areata patients in the past. But a relief after Aclaris’ topical ATI-502 JAK inhibitor failed to treat alopecia areata patients sufficiently well recently.
The latest trial photos are pretty amazing. One of the best ones:
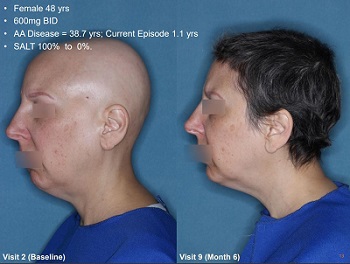
So, does JAK works also in AGA ? If yes why not just try the same JAK used for AA instead of waiting another 10 years of clinical trials for AGA ?
It doesn’t work on AGA. Some patients have both AA and AGA. When they take a JAK inhibitor, their hair grows back from complete baldness into an AGA pattern.
Proof for your statement?
This is my profession. Have seen this exact scenario.
Damn, good for them. It can’t be easy having alopecia areata. I do wish there was something that offered such dramatic results to us with AGA though.
Daniel,
I feel exactly the same way… sincerely happy for the AA patients, but can’t help but be jealous that AGA gets nothing but photos with darker lighting. The day we see photos like this for AGA… can hardly imagine! I just hope it’s soon, brother.
Jonathan- because that is how its done for MPB. Always has to be the most difficult way possible for us haha.
In all seriousness, these are the results all new MPB treatments should be offering us now, not RiverTown crap of 4 new hairs, or all those other treatments offering only minimal results. Full thick dense regrowth of hair results. These are cosmetically appropriate defined regrowth for a 2020 treatment. If follica can produce half of this density then we are in the right direction.
Wow this is great! Happy for AA patients for whom this might become a reality for!
I will also say, the entitlement and negative energy on this board is absurd sometimes. I get disappointment and frustration, and I don’t blame people for feeling this way, however I struggle to understand why there is a need to broadcast such feelings so frequently. Hang in there, we’re all going to make it eventually.
50 percent of negativity is mJones. For almost past 5 years I would guess.
This is really good. Credit to Aclaris. The AA sufferers deserved a cure before the naturally balding amongst us.
I hope the sufferers get easy and cheap access to this cure and any side effects are minimal.
Oral JAK INHIBITOR has not been tested for AGA, only Topical, correct?
This could work well for us.
That percentage is more like 70% negativity and 30% positive messages from me. Come on admin, you should be calculating this better :) Well the only reason why I am negative is because we only get negative results from all these hair loss MPB treatments/companies for the past 20 years. Name me one company that gave us positive great results w/pics that were not altered. We have Follicum 8% leg hair growth, or Rivertown 4 hairs on 78 yr old, or Aderans, or Histogen photoshop Ziereg pics, or breezula results showing drop in maintenance after 6 months or Intercytex failing, Aderans failing, Hairclone asking us for our money to fund their company, or brotzu lotion, aclaris MPB topical results …..I am sure there are many more that I cant think of right now hahhaha that all failed to give positive results.
Just being realistic over here my friend not negative. If you guys find a comb over or 5 hairs of regrowth exciting and positive news then more power to you. Have fun combing that over and hiding the balding.
Although you are always 100 percent positive for Follica and Cotsarelis (your family member I presume)?
Wow. That’s 100% legitimate full regrowth. Now if only this worked for AGA…
Hoping SkinTE pans out because it appears to be the only possible “cure” on the horizon.
Most likely if this product will be marketed it will not cost a fortune… but I’m sure that if something for AGA comes out, they’ll clean up our wallet from every penny…
Admin, HairClone just released their banking service. What does this mean for us? Is thier treatment accessible now? And if so how do we get a hold of it through them?
They need to build a facility before they start injections. They crowd funded for the facility and fell short.
Thus far I have been 50% right since JAK obviously has pretty much 100% worked for AA type hairloss. Back on the limb, I say it will work very well for AGA once they up the dose.
Just wish they would hurry up… but according to Aclaris (and they have been excellent on their timelines) we should know by next summer how well the Higher dose of JAK works on AGA hair loss patients. Thus 9-11 more months to go.
Didn’t it work pretty good considering it was evaluated over a much shorter time period and at a significantly lower dose?
Great results. Just waiting for someone to say that it is the lighting or it’s a combover ;)
Why isn’t this study (previously referenced here) getting more attention?
* A Subset of TREM2+ Dermal Macrophages Secretes Oncostatin M to Maintain Hair Follicle Stem Cell Quiescence and Inhibit Hair Growth *
https://www.cell.com/cell-stem-cell/fulltext/S1934-5909(19)30011-6
If the JAK inhibitors didn’t work well for AGA, then here is a specific biochemical mechanism that “maintains hair follicle stem cell quiescence and inhibits hair growth”.
It seems like that should be a high-priority research target. Find a way to eliminate these “trichophage” cells and solve the problem.
https://www.cuimc.columbia.edu/news/studies-uncover-new-approaches-combat-hair-loss-men-and-women
“Our previous studies implicated JAK-STAT signaling as one potential new therapeutic pathway for hair loss disorders by targeting hair follicle stem cells with JAK inhibitors,” Christiano says. (A biotech company recently reported results of a small phase 2 trial of a topical JAK-STAT inhibitor based on these studies.) “Here, we show that blocking the source of the JAK activating signal outside the hair follicle is another way to target this mechanism.”
Hair bank – https://www.dailymail.co.uk/health/article-7306489/Pioneering-hair-transplant-therapy-gets-green-light-Government-start-freezing-follicles.html
“Meet Paul Kemp. He may be about to announce what for bald men is the Holy Grail – a way of making hair grow again, using stem-cell technology. […] Scientists at Intercytex are trying to create a cell bank […]. Intercytex is looking at robotics to automate the process. In the near future that would mean an answer to the prayers of all those who crave a cure for baldness.”
—Paul Kemp from a 2006(!) article.
https://www.independent.co.uk/news/uk/this-britain/i-have-the-cure-for-baldness-though-it-hasnt-worked-for-me-obviously-6102956.html
I hope Hairclone’s future treatment efficacy has substantially improved from 2006. They seem to anticipate years-out if they are charging a per-year storage fee; less than ten, I’m told. ; P
Ahahahaha
At least, he ain’t a quitter.
Ahahahaha
Our best bet right now is Aclaris’s High Dose JAK results for AGA due next Summer. We see the great photos above. Why Not Us.
Jak is dead. Even if high dose worked it wont be out next summer.
That post makes no sense…on one hand it’s dead and on the other it may or may not work but if it does it won’t be out by next summer. You my friend don’t qualify as a top “stock picker”, your forecasts of the future are too wish washy. If you’re going to make baseless claims go balls out!
Ehhhhh no it’s not. Aga and as are not the same. Keep dreaming
it’s strange, on Aclaris’ photos, people grow their hair back but not their eyebrows …
Apologies for temporarily hijacking the thread but hair freezing is now official for you folks out in the UK looking to preserve your locks for a later age. http://www.ladbible.com/news/technology-uk-approves-treatment-that-freezes-mens-hair-if-theyre-balding-20190802
I’m confused. What’s the point of this when other companies are positioning for the same thing without having you pay for your hairs to be kept in a freezer?
Hairclone states treatment would be available by December 2019 had crowd source funding target been reached. They raised 100k hoping to reach 300k… They have the license ready to go not enough for the facility.
300k…. Think about that. ….that’s just aggravates the living hell out of me. That’s it?! Now they have to go the old venture capital route gonna take another long time ….just pathetic
I’m gonna sip the hateraid with mjones on that one cheers
If it was actually a sure thing the money would be raised in about 35 seconds..
Aclaris stock has taken a beating. I think expectations should be low.
High dose jak inhibitors is not something we should be messing with.
The risk of potential dangerous side effects is very high.
The risk just wont be worth it.
Hairclone is just going to whither away and disappear just like intercytex. Any company asking hair loss sufferers on a blog to invest definitely screams out scam. I never seen any other hair loss company do this. If they had a real cure or great treatment investors would be dumping money right into for their piece of the pie.
Jak topical doesn’t work. Hence the share crash. However, their internal dose is amazing!
Egghead- cheers to some hateraid juice tonight lol.
I do not know about the various issues you brought up. There are a number of reasons that move a stock price. However, we will know next summer if JAK works extremely well for AGA..
I dont think their a scam. Hair multiplication and hair cloning is already possible in Japan us and UK. It’s not a question whether its real its now a question as to building a facility to treat patients. Its not the possibility its the implementation and unfortuanately all of you are correct the proof comes after the largest hurdles have been cleared. The only low hanging fruit is UK and Japans expedited trial process.
Your absolutely right this is 10 years off in america.
Does that mean shiseido, if proven to work, could be delivered fast in the UK??
I think so. Hairclone and shiseido tech sounds very similar. They also both have immediate regulatory access unlike the States.
Prof. Fukuda, Yokohama University … follicle mass production possible with “bead-based hair follicle germ” (bbHFG)”
https://medicalxpress.com/news/2019-07-laboratory-paves-approach-hair-loss.html
Random question. It’s off-topic, but I hope someone can help.
I’m a diffuse thinner. I realise that transplants can only achieve a certain level of density — depending on hair thickness and characteristics etc —and I realise that transplants aren’t ideal for people with diffuse thinning, partly because of shock loss.
So, my question is this: If a diffuse thinner wants to achieve the maximum level of density, does that mean that they could do this with two transplants?
Even it’s works for AGA, and raides the market in 2 years (which will not happen). I would not take it, too dangerous, too unstable.
Hey Paul check out dr. Konior website. He has a new fue procedure that minimizes shockloss. Forgot the name of it something slit and place. You’ll see it on the website. I’m a diffuse thinner also my friend and looking into an fue as well. Have konior, Dr couto in mind. Hoping follica or SM comes out soon to avoid the fue route.
Thanks, Mjones.
Appreciate it!