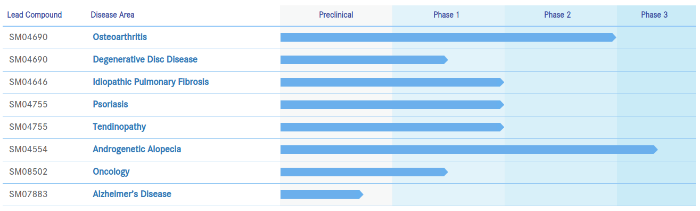
I have covered Samumed (US) close to 20 times on this blog. In 2018, the company started Phase 3 clinical trials for its androgenetic alopecia (SM04554) topical product.
Can Samumed Release Hair Loss Product in 2022?
Yesterday, Samumed’s CEO Dr. Osman Kibar gave an interesting interview to Fierce Biotech (h/t reader “John Doe”). The most relevant part of this interview for us is:
“Its alopecia treatment is close behind, with approval anticipated in late 2021 or early 2022.”
Not a surprise as far as time frame goes, but Mr. Kibar seems confident that they will get approval. I am assuming he agreed with the above sentence in the interview or was quoted as such.
Not sure if other CEOs “anticipate” approval chances before Phase 3 trials are complete. I am hoping that Samumed has seen good results during the first year of ongoing Phase 3 trials in order to warrant this confidence.
The interview also has some interesting information about the scientific reasoning behind why Samumed’s products are safe despite targeting the critical Wnt pathway.
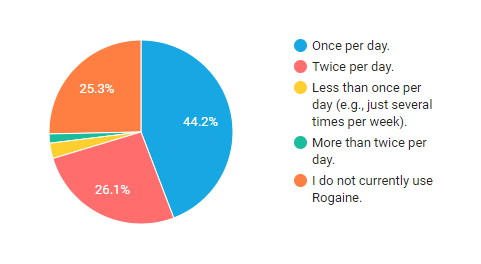
 The instructions on the packaging tell male users to apply Rogaine foam twice per day. However, many people choose to apply it just once per day instead.
The instructions on the packaging tell male users to apply Rogaine foam twice per day. However, many people choose to apply it just once per day instead.